Cardiac sodium channelopathy associated with SCN5A mutations: electrophysiological, molecular and genetic aspects
- PMID: 23818691
- PMCID: PMC3779105
- DOI: 10.1113/jphysiol.2013.256461
Cardiac sodium channelopathy associated with SCN5A mutations: electrophysiological, molecular and genetic aspects
Abstract
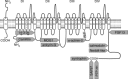
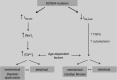
Over the last two decades, an increasing number of SCN5A mutations have been described in patients with long QT syndrome type 3 (LQT3), Brugada syndrome, (progressive) conduction disease, sick sinus syndrome, atrial standstill, atrial fibrillation, dilated cardiomyopathy, and sudden infant death syndrome (SIDS). Combined genetic, electrophysiological and molecular studies have provided insight into the dysfunction and dysregulation of the cardiac sodium channel in the setting of SCN5A mutations identified in patients with these inherited arrhythmia syndromes. However, risk stratification and patient management is hindered by the reduced penetrance and variable disease expressivity in sodium channelopathies. Furthermore, various SCN5A-related arrhythmia syndromes are known to display mixed phenotypes known as cardiac sodium channel overlap syndromes. Determinants of variable disease expressivity, including genetic background and environmental factors, are suspected but still largely unknown. Moreover, it has become increasingly clear that sodium channel function and regulation is more complicated than previously assumed, and the sodium channel may play additional, as of yet unrecognized, roles in cardiac structure and function. Development of cardiac structural abnormalities secondary to SCN5A mutations has been reported, but the clinical relevance and underlying mechanisms are unclear. Increased insight into these issues would enable a major next step in research related to cardiac sodium channel disease, ultimately enabling improved diagnosis, risk stratification and treatment strategies.
Figures


References
-
- Abriel H. Cardiac sodium channel Nav1.5 and interacting proteins: Physiology and pathophysiology. J Mol Cell Cardiol. 2010;48:2–11. - PubMed
-
- Allouis M, Le Bouffant F, Wilders R, Péroz D, Schott JJ, Noireaud J, Le Marec H, Mérot J, Escande D, Baró I. 14-3-3 is a regulator of the cardiac voltage-gated sodium channel Nav1.5. Circ Res. 2006;98:1538–1546. - PubMed
-
- Amin AS, Verkerk AO, Bhuiyan ZA, Wilde AA, Tan HL. Novel Brugada syndrome-causing mutation in ion-conducting pore of cardiac Na+ channel does not affect ion selectivity properties. Acta Physiol Scand. 2005;185:291–301. - PubMed
-
- Antzelevitch C. The Brugada syndrome: diagnostic criteria and cellular mechanisms. Eur Heart J. 2001;22:356–363. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

