The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism
- PMID: 23821742
- PMCID: PMC3735932
- DOI: 10.1194/jlr.R036012
The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism
Abstract
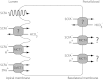
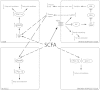
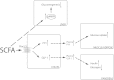
Short-chain fatty acids (SCFAs), the end products of fermentation of dietary fibers by the anaerobic intestinal microbiota, have been shown to exert multiple beneficial effects on mammalian energy metabolism. The mechanisms underlying these effects are the subject of intensive research and encompass the complex interplay between diet, gut microbiota, and host energy metabolism. This review summarizes the role of SCFAs in host energy metabolism, starting from the production by the gut microbiota to the uptake by the host and ending with the effects on host metabolism. There are interesting leads on the underlying molecular mechanisms, but there are also many apparently contradictory results. A coherent understanding of the multilevel network in which SCFAs exert their effects is hampered by the lack of quantitative data on actual fluxes of SCFAs and metabolic processes regulated by SCFAs. In this review we address questions that, when answered, will bring us a great step forward in elucidating the role of SCFAs in mammalian energy metabolism.
Keywords: bacterial short-chain fatty acid metabolism; nutritional fiber; short-chain fatty acid fluxes and concentrations.
Figures

References
-
- Galisteo M., Duarte J., Zarzuelo A. 2008. Effects of dietary fibers on disturbances clustered in the metabolic syndrome. J. Nutr. Biochem. 19: 71–84. - PubMed
-
- Venn B. J., Mann J. I. 2004. Cereal grains, legumes and diabetes. Eur. J. Clin. Nutr. 58: 1443–1461. - PubMed
-
- Delzenne N. M., Cani P. D. 2005. A place for dietary fibre in the management of the metabolic syndrome. Curr. Opin. Clin. Nutr. Metab. Care. 8: 636–640. - PubMed
-
- Marlett J. A., McBurney M. I., Slavin J. L. 2002. Position of the American Dietetic Association: health implications of dietary fiber. J. Am. Diet. Assoc. 102: 993–1000. - PubMed
-
- Nicholson J. K., Holmes E., Kinross J., Burcelin R., Gibson G., Jia W., Pettersson S. 2012. Host-gut microbiota metabolic interactions. Science. 336: 1262–1267. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

