TGF-β-Neutralizing Antibody 1D11 Enhances Cytarabine-Induced Apoptosis in AML Cells in the Bone Marrow Microenvironment
- PMID: 23826077
- PMCID: PMC3695026
- DOI: 10.1371/journal.pone.0062785
TGF-β-Neutralizing Antibody 1D11 Enhances Cytarabine-Induced Apoptosis in AML Cells in the Bone Marrow Microenvironment
Abstract
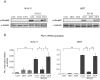
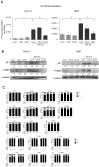
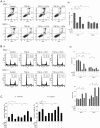
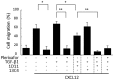
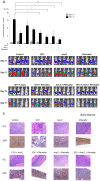
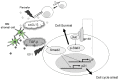
Hypoxia and interactions with bone marrow (BM) stromal cells have emerged as essential components of the leukemic BM microenvironment in promoting leukemia cell survival and chemoresistance. High levels of transforming growth factor beta 1 (TGFβ1) produced by BM stromal cells in the BM niche regulate cell proliferation, survival, and apoptosis, depending on the cellular context. Exogenous TGFβ1 induced accumulation of acute myeloid leukemia (AML) cells in a quiescent G0 state, which was further facilitated by the co-culture with BM-derived mesenchymal stem cells (MSCs). In turn, TGFβ-neutralizing antibody 1D11 abrogated rhTGFβ1 induced cell cycle arrest. Blocking TGFβ with 1D11 further enhanced cytarabine (Ara-C)-induced apoptosis of AML cells in hypoxic and in normoxic conditions. Additional constituents of BM niche, the stroma-secreted chemokine CXCL12 and its receptor CXCR4 play crucial roles in cell migration and stroma/leukemia cell interactions. Treatment with 1D11 combined with CXCR4 antagonist plerixafor and Ara-C decreased leukemia burden and prolonged survival in an in vivo leukemia model. These results indicate that blockade of TGFβ by 1D11 and abrogation of CXCL12/CXCR4 signaling may enhance the efficacy of chemotherapy against AML cells in the hypoxic BM microenvironment.
Conflict of interest statement
Figures
Similar articles
-
TGF-β1 and CXCL12 modulate proliferation and chemotherapy sensitivity of acute myeloid leukemia cells co-cultured with multipotent mesenchymal stromal cells.Hematology. 2018 Jul;23(6):337-345. doi: 10.1080/10245332.2017.1402455. Epub 2017 Nov 15. Hematology. 2018. PMID: 29140182
-
Plerixafor as a chemosensitizing agent in pediatric acute lymphoblastic leukemia: efficacy and potential mechanisms of resistance to CXCR4 inhibition.Oncotarget. 2014 Oct 15;5(19):8947-58. doi: 10.18632/oncotarget.2407. Oncotarget. 2014. PMID: 25333254 Free PMC article.
-
Defining the in vivo characteristics of acute myeloid leukemia cells behavior by intravital imaging.Immunol Cell Biol. 2019 Feb;97(2):229-235. doi: 10.1111/imcb.12216. Epub 2018 Dec 13. Immunol Cell Biol. 2019. PMID: 30422351 Free PMC article.
-
Dissecting the role of the CXCL12/CXCR4 axis in acute myeloid leukaemia.Br J Haematol. 2020 Jun;189(5):815-825. doi: 10.1111/bjh.16456. Epub 2020 Mar 5. Br J Haematol. 2020. PMID: 32135579 Review.
-
Role of CXCR4 in the pathogenesis of acute myeloid leukemia.Theranostics. 2013;3(1):34-9. doi: 10.7150/thno.5150. Epub 2013 Jan 13. Theranostics. 2013. PMID: 23382784 Free PMC article. Review.
Cited by
-
Limb-Bud and Heart (LBH) Upregulation in Cardiomyocytes under Hypoxia Promotes the Activation of Cardiac Fibroblasts via Exosome Secretion.Mediators Inflamm. 2022 Sep 6;2022:8939449. doi: 10.1155/2022/8939449. eCollection 2022. Mediators Inflamm. 2022. PMID: 36110098 Free PMC article.
-
Inflammation as a driver of hematological malignancies.Front Oncol. 2024 Mar 20;14:1347402. doi: 10.3389/fonc.2024.1347402. eCollection 2024. Front Oncol. 2024. PMID: 38571491 Free PMC article. Review.
-
Targeting the tumor microenvironment to enhance antitumor immune responses.Oncotarget. 2015 Jan 30;6(3):1359-81. doi: 10.18632/oncotarget.3204. Oncotarget. 2015. PMID: 25682197 Free PMC article. Review.
-
Preventing and Repairing Myeloma Bone Disease by Combining Conventional Antiresorptive Treatment With a Bone Anabolic Agent in Murine Models.J Bone Miner Res. 2019 May;34(5):783-796. doi: 10.1002/jbmr.3606. Epub 2018 Nov 26. J Bone Miner Res. 2019. PMID: 30320927 Free PMC article.
-
Azacitidine combined with the selective FLT3 kinase inhibitor crenolanib disrupts stromal protection and inhibits expansion of residual leukemia-initiating cells in FLT3-ITD AML with concurrent epigenetic mutations.Oncotarget. 2017 Oct 16;8(65):108738-108759. doi: 10.18632/oncotarget.21877. eCollection 2017 Dec 12. Oncotarget. 2017. PMID: 29312564 Free PMC article.
References
-
- Garrido SM, Appelbaum FR, Willman CL, Banker DE (2001) Acute myeloid leukemia cells are protected from spontaneous and drug-induced apoptosis by direct contact with a human bone marrow stromal cell line (HS-5). Experimental Hematology 29: 448–457. - PubMed
-
- Taichman RS (2005) Blood and bone: two tissues whose fates are intertwined to create the hematopoietic stem-cell niche. Blood 105: 2631–2639. - PubMed
-
- Jang CW, Chen CH, Chen CC, Chen JY, Su YH (2002) TGF-beta induces apoptosis through Smad-mediated expression of DAP-kinase. Nature Cell Biol 4: 51–58. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

