Lack of Plasma Protein Hemopexin Results in Increased Duodenal Iron Uptake
- PMID: 23826373
- PMCID: PMC3694894
- DOI: 10.1371/journal.pone.0068146
Lack of Plasma Protein Hemopexin Results in Increased Duodenal Iron Uptake
Abstract
Purpose: The body concentration of iron is regulated by a fine equilibrium between absorption and losses of iron. Iron can be absorbed from diet as inorganic iron or as heme. Hemopexin is an acute phase protein that limits iron access to microorganisms. Moreover, it is the plasma protein with the highest binding affinity for heme and thus it mediates heme-iron recycling. Considering its involvement in iron homeostasis, it was postulated that hemopexin may play a role in the physiological absorption of inorganic iron.
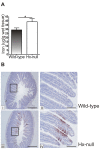
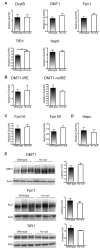
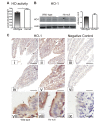
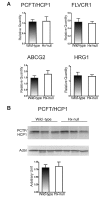
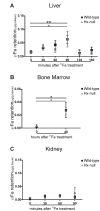
Methods and results: Hemopexin-null mice showed elevated iron deposits in enterocytes, associated with higher duodenal H-Ferritin levels and a significant increase in duodenal expression and activity of heme oxygenase. The expression of heme-iron and inorganic iron transporters was normal. The rate of iron absorption was assessed by measuring the amount of (57)Fe retained in tissues from hemopexin-null and wild-type animals after administration of an oral dose of (57)FeSO4 or of (57)Fe-labelled heme. Higher iron retention in the duodenum of hemopexin-null mice was observed as compared with normal mice. Conversely, iron transfer from enterocytes to liver and bone marrow was unaffected in hemopexin-null mice.
Conclusions: The increased iron level in hemopexin-null duodenum can be accounted for by an increased iron uptake by enterocytes and storage in ferritins. These data indicate that the lack of hemopexin under physiological conditions leads to an enhanced duodenal iron uptake thus providing new insights to our understanding of body iron homeostasis.
Conflict of interest statement
Figures


References
-
- Dunn LL, Rahmanto YS, Richardson DR (2007) Iron uptake and metabolism in the new millennium. Trends Cell Biol 17: 93-100. doi:10.1016/j.tcb.2006.12.003. PubMed: 17194590. - DOI - PubMed
-
- Krishnamurthy P, Schuetz JD (2006) Role of ABCG2/BCRP in biology and medicine. Annu Rev Pharmacol Toxicol 46: 381-410. doi:10.1146/annurev.pharmtox.46.120604.141238. PubMed: 16402910. - DOI - PubMed
-
- Keel SB, Doty RT, Yang Z, Quigley JG, Chen J et al. (2008) A heme export protein is required for red blood cell differentiation and iron homeostasis. Science 319: 825-828. doi:10.1126/science.1151133. PubMed: 18258918. - DOI - PubMed
-
- Chiabrando D, Marro S, Mercurio S, Giorgi C, Petrillo S et al. (2012) The mitochondrial heme exporter FLVCR1b mediates erythroid differentiation. J Clin Invest 122: 4569-4579. doi:10.1172/JCI62422. PubMed: 23187127. - DOI - PMC - PubMed
-
- Knutson MD (2010) Iron-sensing proteins that regulate hepcidin and enteric iron absorption. Annu Rev Nutr 30: 149-171. doi:10.1146/annurev.nutr.012809.104801. PubMed: 20415583. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

