Mice deficient in the St3gal3 gene product α2,3 sialyltransferase (ST3Gal-III) exhibit enhanced allergic eosinophilic airway inflammation
- PMID: 23830412
- PMCID: PMC3874253
- DOI: 10.1016/j.jaci.2013.05.018
Mice deficient in the St3gal3 gene product α2,3 sialyltransferase (ST3Gal-III) exhibit enhanced allergic eosinophilic airway inflammation
Abstract
Background: Sialic acid-binding immunoglobulin-like lectin (Siglec)-F is a proapoptotic receptor on mouse eosinophils, but little is known about its natural tissue ligand.
Objective: We previously reported that the St3gal3 gene product α2,3 sialyltransferase (ST3Gal-III) is required for constitutive Siglec-F lung ligand synthesis. We therefore hypothesized that attenuation of ST3Gal-III will decrease Siglec-F ligand levels and enhance allergic eosinophilic airway inflammation.
Methods: C57BL/6 wild-type mice and St3gal3 heterozygous or homozygous deficient (St3gal3(+/-) and St3gal3(-/-)) mice were used. Eosinophilic airway inflammation was induced through sensitization to ovalbumin (OVA) and repeated airway OVA challenge. Siglec-F human IgG1 fusion protein (Siglec-F-Fc) was used to detect Siglec-F ligands. Lung tissue and bronchoalveolar lavage fluid (BALF) were analyzed for inflammation, as well as various cytokines and chemokines. Serum was analyzed for allergen-specific immunoglobulin levels.
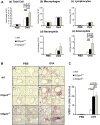
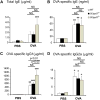
Results: Western blotting with Siglec-F-Fc detected approximately 500-kDa and approximately 200-kDa candidate Siglec-F ligands that were less abundant in St3gal3(+/-) lung extracts and nearly absent in St3gal3(-/-) lung extracts. After OVA sensitization and challenge, Siglec-F ligands were increased in wild-type mouse lungs but less so in St3gal3 mutants, whereas peribronchial and BALF eosinophil numbers were greater in the mutants, with the following rank order: St3gal3(-/-) ≥ St3gal3(+/-) > wild-type mice. Levels of various cytokines and chemokines in BALF were not significantly different among these 3 types of mice, although OVA-specific serum IgG1 levels were increased in St3gal3(-/-) mice.
Conclusions: After OVA sensitization and challenge, St3gal3(+/-) and St3gal3(-/-) mice have more intense allergic eosinophilic airway inflammation and less sialylated Siglec-F ligands in their airways. One possible explanation for these findings is that levels of sialylated airway ligands for Siglec-F might be diminished in mice with attenuated levels of ST3Gal-III, resulting in a reduction in a natural proapoptotic pathway for controlling airway eosinophilia.
Keywords: 6’-su-sLacNAc; 6′-Sulfated sialyl Lewis X; 6′-Sulfated sialyl N-acetyl-D-lactosamine; 6′-su-sLe(x); 6′-sulfated sialyl Lewis X; 6′-sulfated sialyl N-acetyl-D-lactosamine; BALF; Bronchoalveolar lavage fluid; Eosinophils; MBP; Major basic protein; OVA; Ovalbumin; ST3Gal-III; Sialic acid–binding immunoglobulin-like lectin; Siglec; Siglec-8 human IgG(1) Fc chimera; Siglec-8-Fc; Siglec-F; Siglec-F human IgG(1) Fc chimera; Siglec-F-Fc; St3gal3; St3gal3 gene product α2,3 sialyltransferase; WT; Wild-type; apoptosis; asthma; glycan ligands; lung.
Copyright © 2013 American Academy of Allergy, Asthma & Immunology. Published by Mosby, Inc. All rights reserved.
Conflict of interest statement
Disclosure of potential conflict of interest: T. Kiwamoto, F. Wu, M. G. Motari, and Z. Zhu have received grants from the National Institutes of Health (NIH). D. F Smith has received a grant from Emory University School of Medicine. R. L. Schnaar has received grants and travel support from the NIH, has received royalties from Oxford University Press and Johns Hopkins, has stock/stock options in Zacharon Pharmaceuticals, and has received travel support from Academia Sinica and the University of Alberta. B. S. Bochner has received a grant from the NIH, has consultant arrangements with Allakos, has patents through Johns Hopkins University regarding Siglec-8 and its ligand, receives royalties from Johns Hopkins University, and has an equity position in Allakos. M. E. Brummet declares that she has no relevant conflicts of interest.
Figures


Similar articles
-
Sialyltransferase ST3Gal-III regulates Siglec-F ligand formation and eosinophilic lung inflammation in mice.J Immunol. 2013 Jun 15;190(12):5939-48. doi: 10.4049/jimmunol.1203455. Epub 2013 May 15. J Immunol. 2013. PMID: 23677475 Free PMC article.
-
Characterization of expression of glycan ligands for Siglec-F in normal mouse lungs.Am J Respir Cell Mol Biol. 2011 Feb;44(2):238-43. doi: 10.1165/rcmb.2010-0007OC. Epub 2010 Apr 15. Am J Respir Cell Mol Biol. 2011. PMID: 20395633 Free PMC article.
-
Airway glycomic and allergic inflammatory consequences resulting from keratan sulfate galactose 6-O-sulfotransferase (CHST1) deficiency.Glycobiology. 2018 Jun 1;28(6):406-417. doi: 10.1093/glycob/cwy025. Glycobiology. 2018. PMID: 29659839 Free PMC article.
-
The role of lung epithelial ligands for Siglec-8 and Siglec-F in eosinophilic inflammation.Curr Opin Allergy Clin Immunol. 2013 Feb;13(1):106-11. doi: 10.1097/ACI.0b013e32835b594a. Curr Opin Allergy Clin Immunol. 2013. PMID: 23160308 Free PMC article. Review.
-
Siglec-8 on human eosinophils and mast cells, and Siglec-F on murine eosinophils, are functionally related inhibitory receptors.Clin Exp Allergy. 2009 Mar;39(3):317-24. doi: 10.1111/j.1365-2222.2008.03173.x. Clin Exp Allergy. 2009. PMID: 19178537 Free PMC article. Review.
Cited by
-
Siglec-mediated regulation of immune cell function in disease.Nat Rev Immunol. 2014 Oct;14(10):653-66. doi: 10.1038/nri3737. Epub 2014 Sep 19. Nat Rev Immunol. 2014. PMID: 25234143 Free PMC article. Review.
-
Glycobiology simplified: diverse roles of glycan recognition in inflammation.J Leukoc Biol. 2016 Jun;99(6):825-38. doi: 10.1189/jlb.3RI0116-021R. Epub 2016 Mar 22. J Leukoc Biol. 2016. PMID: 27004978 Free PMC article. Review.
-
Sialic acid in the regulation of blood cell production, differentiation and turnover.Immunology. 2024 Aug;172(4):517-532. doi: 10.1111/imm.13780. Epub 2024 Mar 19. Immunology. 2024. PMID: 38503445 Free PMC article. Review.
-
Has2 Regulates the Development of Ovalbumin-Induced Airway Remodeling and Steroid Insensitivity in Mice.Front Immunol. 2022 Jan 7;12:770305. doi: 10.3389/fimmu.2021.770305. eCollection 2021. Front Immunol. 2022. PMID: 35069543 Free PMC article.
-
Systematic Chemoenzymatic Synthesis of O-Sulfated Sialyl Lewis x Antigens.Chem Sci. 2016 Apr 1;7(4):2827-2831. doi: 10.1039/C5SC04104J. Epub 2015 Dec 17. Chem Sci. 2016. PMID: 28138383 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

