A Single Gradient Stability-Indicating Reversed-Phase LC Method for the Estimation of Impurities in Omeprazole and Domperidone Capsules
- PMID: 23833712
- PMCID: PMC3700074
- DOI: 10.3797/scipharm.1209-12
A Single Gradient Stability-Indicating Reversed-Phase LC Method for the Estimation of Impurities in Omeprazole and Domperidone Capsules
Abstract
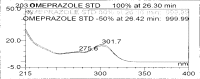
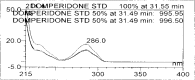
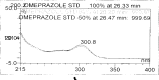
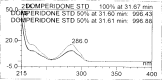
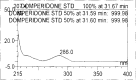
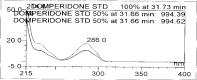
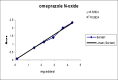
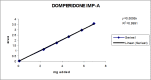
A gradient reversed-phase liquid chromatographic (RP-LC) method was developed for the quantitative estimation of impurities in the pharmaceutical dosage form of Omeprazole and Domperidone capsules. The developed method is a stability-indicating test method for the estimation of impurities generated during the formulation and storage of Omeprazole and Domperidone capsules. The chromatographic separation was achieved on a column packed with octadecyl silane, having a column length of 250 mm and diameter of 4.6 mm with a particle size of 5 μm, and by following a gradient program using a combination of a monobasic potassium phosphate buffer (0.05M) and acetonitrile. Since the spectral properties were similar, both compounds' individual impurities were estimated at 285 nm. Forced degradation studies were performed on Omeprazole pellets (enteric coated) and Domperidone pellets (SR coated) encapsulated in size '1' hard gelatin capsules. Omeprazole and Domperidone were degraded using acid hydrolysis (0.1 N hydrochloric acid), base (0.1 N sodium hydroxide), oxidation (50% hydrogen peroxide), heat (105 °C), and UV light (254 nm). The established method was validated and found to be linear, accurate, precise, specific, robust, and rugged.
Keywords: Domperidone; Forced Degradation; Method validation; Omeprazole; RP-LC.
Figures






















References
-
- Aydan Y, Rustii O. Specific H+/K+-ATPase inhibitors decreased contractile responses of isolated rat vas deferens. Pharmacol Res. 2006;54:397–405. http://dx.doi.org/10.1016/j.phrs.2006.07.005. - DOI - PubMed
-
- Shimatani T, Inoue M, Kuroiwa T, Xu J, Mieno H, Tazuma S. Acid suppressive effect of Generic Omeprazole, comparison of 3 brands of Generics with Omeprazole. Dig Liver Dis. 2006;38:554–559. http://dx.doi.org/10.1016/j.dld.2006.01.032. - DOI - PubMed
-
- Ahmad N, Keith-Ferris J, Gooden E, Abell T. Making a case for Domperidone in the treatment of gastro intestinal motility disorders. Curr Opin Pharmacol. 2006;6:571–576. http://dx.doi.org/10.1016/j.coph.2006.07.004. - DOI - PubMed
-
- Drug Bank Mechanism of action of Domperidone. Domperidone; Accession Number: APRD00418.
-
- Naser LR, Keven CB, Angela DMK. A simple sensitive bioanalytical assay for simultaneous determination of omeprazole and its 3 major metabolites in human blood plasma using RP-HPLC after a simple liquid-liquid extraction procedure. J Chromatogr B. 2006;844:314–321. http://dx.doi.org/10.1016/j.jchromb.2006.07.047. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
