Comparative study analyzing survival and safety of bevacizumab/carboplatin/paclitaxel and cisplatin/pemetrexed in chemotherapy-naïve patients with advanced non-squamous bronchogenic carcinoma not harboring EGFR mutation
- PMID: 23836994
- PMCID: PMC3702548
- DOI: 10.2147/OTT.S45906
Comparative study analyzing survival and safety of bevacizumab/carboplatin/paclitaxel and cisplatin/pemetrexed in chemotherapy-naïve patients with advanced non-squamous bronchogenic carcinoma not harboring EGFR mutation
Abstract
Purpose: The majority of Egyptian patients with lung cancer present at a late stage of the disease. Bevacizumab/carboplatin/paclitaxel, as well as cisplatin plus pemetrexed, are both standard regimens for advanced non-squamous bronchogenic cancer. This study compares both regimens, in terms of efficacy and toxicity profile, in Egyptian patients.
Patients and methods: This is a randomized Phase II study comparing toxicity profile and survival in 41 chemotherapy-naïve patients with stage IIIB or IV non-squamous NSCLC, with an ECOG performance status of 0 to 2. The epidermal growth factor receptor (EGFR) mutation detection was performed prior to treatment of all patients. Patients in the first group received: bevacizumab 7.5 mg/m(2) on Day 1 and Day 15; carboplatin area under the curve-5 on Day 1; and paclitaxel 60 mg/m(2) on Day 1, Day 8, and Day 15 every 4 weeks. In the second group, patients received cisplatin 75 mg/m(2) and pemetrexed 500 mg/m(2) every 3 weeks.
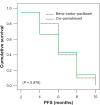
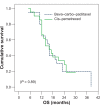
Results: The combination of bevacizumab/carboplatin/paclitaxel demonstrated higher Grade III-IV toxicity than cisplatin/pemetrexed regarding sensory/motor neuropathy (P = 0.06), DVT (P = 0.23), proteinuria (P = 0.23), and hypertension (P = 0.11), as well as Grade II alopecia (P = 0.001); however, no significant difference in toxicities between both arms was recorded regarding nausea and vomiting (P = 0.66), hematological toxicity, febrile neutropenia (P = 1) and fatigue (P = 0.66). Progression-free survival was similar for both treatment arms with a median of 6 months (P = 0.978). Overall median survival was comparable in both arms, 16.07 months versus 16.01 months (P = 0.89).
Conclusion: Bevacizumab/carboplatin/paclitaxel and cisplatin/pemetrexed provided meaningful and comparable efficacy in advanced non-squamous bronchogenic carcinoma not harboring EGFR mutation. No significant difference in toxicity was observed between both treatment arms, apart from bevacizumab/carboplatin/paclitaxel-related risks as DVT, hypertension, proteinuria, sensory/motor neuropathy, and alopecia.
Keywords: NSCLC; bevacizumab; non-small cell lung cancer; pemetrexed.
Figures


References
-
- Karim-Kos HE, de Vries E, Soerjomataram I, Lemmens V, Siesling S, Coebergh JW. Recent trends of cancer in Europe: a combined approach of incidence, survival, and mortality for 17 cancer sites since the 1990s. Eur J Cancer. 2008;44(10):1345–1389. - PubMed
-
- Govindan R, Page N, Morgensztern D, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol. 2006;24(28):4539–4544. - PubMed
-
- Pfister DG, Johnson DH, Azzoli CG, et al. American Society of Clinical Oncology treatment of unresectable non-small-cell lung cancer guideline: update 2003. J Clin Oncol. 2004;22(2):330–353. - PubMed
-
- Schiller JH, Harrington D, Belani CP, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med. 2002;346(2):92–98. - PubMed
-
- Scagliotti GV, De Marinis F, Rinaldi M, et al. Phase III randomized trial comparing three platinum-based doublets in advanced non-small-cell lung cancer. J Clin Oncol. 2002;20(21):4285–4291. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

