The molecular and biophysical characterization of the human blood-nerve barrier: current concepts
- PMID: 23839247
- PMCID: PMC4030640
- DOI: 10.1159/000353293
The molecular and biophysical characterization of the human blood-nerve barrier: current concepts
Abstract
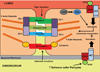
The internal microenvironment in peripheral nerves is highly regulated in order to maintain normal axonal impulse transmission to or from the central nervous system. In humans, this regulation is facilitated by specialized tight junction (TJ)-forming endoneurial microvascular endothelial cells and perineurial myofibroblasts that form multiple concentric layers around nerve fascicles. The endoneurial endothelial cells come in direct contact with circulating blood and, thus, can be considered the blood-nerve barrier (BNB). Studies on the molecular and biophysical properties of the human BNB in vivo or in situ are limited. Owing to the recent isolation of primary human endoneurial endothelial cells and the development of simian virus 40 large T-antigen immortalized cell lines, data are emerging on the structural and functional characteristics of these cells. These data aim to increase our understanding of how solutes, macromolecules, nutrients and hematogenous leukocytes gain access into or are restricted from the endoneurium of peripheral nerves. These concepts have clinical relevance in understanding normal peripheral nerve homeostasis, the response of peripheral nerves to external insult and stresses such as drugs and toxins and the pathogenesis of peripheral neuropathies. This review discusses current knowledge in this nascent and exciting field of microvascular biology.
Copyright © 2013 S. Karger AG, Basel.
Figures



References
-
- Olsson Y. Microenvironment of the peripheral nervous system under normal andpathological conditions. Crit Rev Neurobiol. 1990;5:265–311. - PubMed
-
- Reina M, López A, Villanueva M, de Andrés J, León G. [morphology of peripheralnerves, their sheaths, and their vascularization] Rev Esp Anestesiol Reanim. 2000;47:464–475. - PubMed
-
- Reina M, López A, Villanueva M, De Andrés J, Machés F. [the blood-nerve barrier inperipheral nerves] Rev Esp Anestesiol Reanim. 2003;50:80–86. - PubMed
-
- Bell M, Weddell A. A descriptive study of the blood vessels of the sciatic nerve in therat, man and other mammals. Brain. 1984;107 ( Pt 3):871–898. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

