ATP consumption by sarcoplasmic reticulum Ca²⁺ pumps accounts for 40-50% of resting metabolic rate in mouse fast and slow twitch skeletal muscle
- PMID: 23840903
- PMCID: PMC3698183
- DOI: 10.1371/journal.pone.0068924
ATP consumption by sarcoplasmic reticulum Ca²⁺ pumps accounts for 40-50% of resting metabolic rate in mouse fast and slow twitch skeletal muscle
Abstract
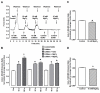
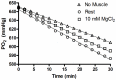
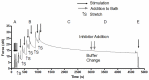
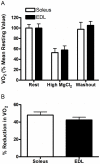
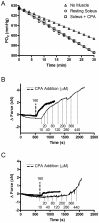
The main purpose of this study was to directly quantify the relative contribution of Ca²⁺ cycling to resting metabolic rate in mouse fast (extensor digitorum longus, EDL) and slow (soleus) twitch skeletal muscle. Resting oxygen consumption of isolated muscles (VO₂, µL/g wet weight/s) measured polarographically at 30°C was ~20% higher (P<0.05) in soleus (0.326 ± 0.022) than in EDL (0.261 ± 0.020). In order to quantify the specific contribution of Ca²⁺ cycling to resting metabolic rate, the concentration of MgCl₂ in the bath was increased to 10 mM to block Ca²⁺ release through the ryanodine receptor, thus eliminating a major source of Ca²⁺ leak from the sarcoplasmic reticulum (SR), and thereby indirectly inhibiting the activity of the sarco(endo) plasmic reticulum Ca²⁺-ATPases (SERCAs). The relative (%) reduction in muscle VO₂ in response to 10 mM MgCl₂ was similar between soleus (48.0±3.7) and EDL (42.4±3.2). Using a different approach, we attempted to directly inhibit SERCA ATPase activity in stretched EDL and soleus muscles (1.42x optimum length) using the specific SERCA inhibitor cyclopiazonic acid (CPA, up to 160 µM), but were unsuccessful in removing the energetic cost of Ca²⁺ cycling in resting isolated muscles. The results of the MgCl₂ experiments indicate that ATP consumption by SERCAs is responsible for 40-50% of resting metabolic rate in both mouse fast- and slow-twitch muscles at 30°C, or 12-15% of whole body resting VO₂. Thus, SERCA pumps in skeletal muscle could represent an important control point for energy balance regulation and a potential target for metabolic alterations to oppose obesity.
Conflict of interest statement
Figures


References
-
- Zurlo F, Larson K, Bogardus C, Ravussin E (1990) Skeletal muscle metabolism is a major determinant of resting energy expenditure. J Clin Invest 86: 1423-1427. doi:10.1172/JCI114857. PubMed: 2243122. - DOI - PMC - PubMed
-
- Toyoshima C (2008) Structural aspects of ion pumping by Ca2+-ATPase of sarcoplasmic reticulum. Arch Biochem Biophys 476: 3-11. doi:10.1016/j.abb.2008.04.017. PubMed: 18455499. - DOI - PubMed
-
- Inesi G, Kurzmack M, Verjovski-Almeida S (1978) ATPase phosphorylation and calcium ion translocation in the transient state of sarcoplasmic reticulum activity. Ann N Y Acad Sci 307: 224-227. doi:10.1111/j.1749-6632.1978.tb41947.x. PubMed: 152088. - DOI - PubMed
-
- de Meis L (2001) Uncoupled ATPase activity and heat production by the sarcoplasmic reticulum Ca2+-ATPase: Regulation by ADP. J Biol Chem 276: 25078-25087. doi:10.1074/jbc.M103318200. PubMed: 11342561. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

