Spinal μ and δ opioids inhibit both thermal and mechanical pain in rats
- PMID: 23843537
- PMCID: PMC3855450
- DOI: 10.1523/JNEUROSCI.1631-13.2013
Spinal μ and δ opioids inhibit both thermal and mechanical pain in rats
Abstract
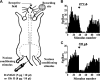
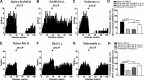
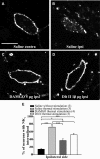
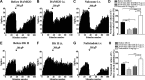
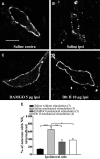
The expression and contribution of μ (MOPR) and δ opioid receptors (DOPR) in polymodal nociceptors have been recently challenged. Indeed, MOPR and DOPR were shown to be expressed in distinct subpopulation of nociceptors where they inhibit pain induced by noxious heat and mechanical stimuli, respectively. In the present study, we used electrophysiological measurements to assess the effect of spinal MOPR and DOPR activation on heat-induced and mechanically induced diffuse noxious inhibitory controls (DNICs). We recorded from wide dynamic range neurons in the spinal trigeminal nucleus of anesthetized rats. Trains of 105 electrical shocks were delivered to the excitatory cutaneous receptive field. DNICs were triggered either by immersion of the hindpaw in 49°C water or application of 300 g of mechanical pressure. To study the involvement of peptidergic primary afferents in the activation of DNIC by noxious heat and mechanical stimulations, substance P release was measured in the spinal cord by visualizing neurokinin type 1 receptor internalization. We found that the activation of spinal MOPR and DOPR similarly attenuates the DNIC and neurokinin type 1 receptor internalization induced either by heat or mechanical stimuli. Our results therefore reveal that the activation of spinal MOPR and DOPR relieves both heat-induced and mechanically induced pain with similar potency and suggest that these receptors are expressed on polymodal, substance P-expressing neurons.
Figures

Similar articles
-
Activation of spinal mu- and delta-opioid receptors potently inhibits substance P release induced by peripheral noxious stimuli.J Neurosci. 2011 Sep 14;31(37):13068-77. doi: 10.1523/JNEUROSCI.1817-11.2011. J Neurosci. 2011. PMID: 21917790 Free PMC article.
-
Contribution of spinal inhibitory receptors in heterosegmental antinociception induced by noxious stimulation.Eur J Neurosci. 2003 Dec;18(11):2999-3006. doi: 10.1111/j.1460-9568.2003.03031.x. Eur J Neurosci. 2003. PMID: 14656295
-
Induction of delta-opioid receptor function in the midbrain after chronic morphine treatment.J Neurosci. 2005 Mar 23;25(12):3192-8. doi: 10.1523/JNEUROSCI.4585-04.2005. J Neurosci. 2005. PMID: 15788776 Free PMC article.
-
Aiming at Ideal Therapeutics-MOPr/DOPr or MOPr-DOPr Heteromertargeting Ligand.Curr Top Med Chem. 2020;20(31):2843-2851. doi: 10.2174/1568026620666200423095231. Curr Top Med Chem. 2020. PMID: 32324516 Review.
-
The activation of bulbo-spinal controls by peripheral nociceptive inputs: diffuse noxious inhibitory controls.Biol Res. 1995;28(1):113-25. Biol Res. 1995. PMID: 8728826 Review.
Cited by
-
Functional Divergence of Delta and Mu Opioid Receptor Organization in CNS Pain Circuits.Neuron. 2018 Apr 4;98(1):90-108.e5. doi: 10.1016/j.neuron.2018.03.002. Epub 2018 Mar 22. Neuron. 2018. PMID: 29576387 Free PMC article.
-
Opioid receptor trafficking and interaction in nociceptors.Br J Pharmacol. 2015 Jan;172(2):364-74. doi: 10.1111/bph.12653. Epub 2014 Jul 1. Br J Pharmacol. 2015. PMID: 24611685 Free PMC article. Review.
-
Developments in Understanding Diffuse Noxious Inhibitory Controls: Pharmacological Evidence from Pre-Clinical Research.J Pain Res. 2021 Apr 20;14:1083-1095. doi: 10.2147/JPR.S258602. eCollection 2021. J Pain Res. 2021. PMID: 33907456 Free PMC article. Review.
-
Alleviating pain with delta opioid receptor agonists: evidence from experimental models.J Neural Transm (Vienna). 2020 Apr;127(4):661-672. doi: 10.1007/s00702-020-02172-4. Epub 2020 Mar 18. J Neural Transm (Vienna). 2020. PMID: 32189076 Review.
-
Berberine Improves Intestinal Motility and Visceral Pain in the Mouse Models Mimicking Diarrhea-Predominant Irritable Bowel Syndrome (IBS-D) Symptoms in an Opioid-Receptor Dependent Manner.PLoS One. 2015 Dec 23;10(12):e0145556. doi: 10.1371/journal.pone.0145556. eCollection 2015. PLoS One. 2015. PMID: 26700862 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
