Mild and efficient cyanuric chloride catalyzed Pictet-Spengler reaction
- PMID: 23843919
- PMCID: PMC3701375
- DOI: 10.3762/bjoc.9.140
Mild and efficient cyanuric chloride catalyzed Pictet-Spengler reaction
Abstract
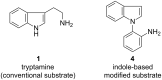
A practical, mild and efficient protocol for the Pictet-Spengler reaction catalyzed by cyanuric chloride (trichloro-1,3,5-triazine, TCT) is described. The 6-endo cyclization of tryptophan/tryptamine and modified Pictet-Spengler substrates with both electron-withdrawing and electron-donating aldehydes was carried out by using a catalytic amount of TCT (10 mol %) in DMSO under a nitrogen atmosphere. TCT catalyzed the Pictet-Spengler reaction involving electron-donating aldehydes in excellent yield. Thus, it has a distinct advantage over the existing methodologies where electron-donating aldehydes failed to undergo 6-endo cyclization. Our methodology provided broad substrate scope and diversity. This is indeed the first report of the use of TCT as a catalyst for the Pictet-Spengler reaction.
Keywords: 6-endo cyclization; Pictet–Spengler; TCT; cyanuric chloride; β-carboline.
Figures
Similar articles
-
Exploring the Chemistry of Spiroindolenines by Mechanistically-Driven Reaction Development: Asymmetric Pictet-Spengler-type Reactions and Beyond.Acc Chem Res. 2020 Apr 21;53(4):974-987. doi: 10.1021/acs.accounts.0c00074. Epub 2020 Apr 10. Acc Chem Res. 2020. PMID: 32275392
-
Regioselective, unconventional Pictet-Spengler cyclization strategy toward the synthesis of benzimidazole-linked imidazoquinoxalines on a soluble polymer support.Chem Asian J. 2011 Jun 6;6(6):1557-65. doi: 10.1002/asia.201000913. Epub 2011 Apr 5. Chem Asian J. 2011. PMID: 21472995
-
Gold-catalyzed domino cycloisomerization/Pictet-Spengler reaction of 2-(4-aminobut-1-yn-1-yl)anilines with aldehydes: synthesis of tetrahydropyrido[4,3-b]indole scaffolds.J Org Chem. 2012 Dec 21;77(24):11355-61. doi: 10.1021/jo302068e. Epub 2012 Dec 13. J Org Chem. 2012. PMID: 23205599
-
The Chiral Pool in the Pictet-Spengler Reaction for the Synthesis of β-Carbolines.Molecules. 2016 May 27;21(6):699. doi: 10.3390/molecules21060699. Molecules. 2016. PMID: 27240334 Free PMC article. Review.
-
Application of Pictet-Spengler Reaction to Indole-Based Alkaloids Containing Tetrahydro-β-carboline Scaffold in Combinatorial Chemistry.ACS Comb Sci. 2017 Apr 10;19(4):199-228. doi: 10.1021/acscombsci.6b00184. Epub 2017 Mar 17. ACS Comb Sci. 2017. PMID: 28276678 Review.
Cited by
-
The Pictet-Spengler Reaction Updates Its Habits.Molecules. 2020 Jan 19;25(2):414. doi: 10.3390/molecules25020414. Molecules. 2020. PMID: 31963860 Free PMC article. Review.
-
Synthesis and Investigation of Tetrahydro-β-carboline Derivatives as Inhibitors of Plant Pathogenic Fungi.Molecules. 2021 Jan 3;26(1):207. doi: 10.3390/molecules26010207. Molecules. 2021. PMID: 33401587 Free PMC article.
References
-
- Cox E D, Cook J M. Chem Rev. 1995;95:1797–1842. doi: 10.1021/cr00038a004. and references therein. - DOI
-
- Nielsen T E, Diness F, Meldal M. Curr Opin Drug Discovery Dev. 2003;6:801–814. - PubMed
-
- Youn S W. Org Prep Proced Int. 2006;38:505–591. doi: 10.1080/00304940609356447. - DOI
-
- Larghi E L, Amongero M, Bracca A B J, Kaufman T S. ARKIVOC. 2005:98–153. doi: 10.3998/ark.5550190.0006.c09. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources