Amyloid-β protofibrils: size, morphology and synaptotoxicity of an engineered mimic
- PMID: 23843949
- PMCID: PMC3699592
- DOI: 10.1371/journal.pone.0066101
Amyloid-β protofibrils: size, morphology and synaptotoxicity of an engineered mimic
Erratum in
- PLoS One. 2013;8(10). doi:10.1371/annotation/44be8a39-d943-419b-a430-c2b30dafadec
Abstract
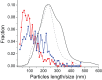
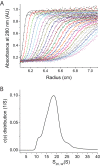
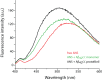
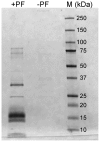
Structural and biochemical studies of the aggregation of the amyloid-β peptide (Aβ) are important to understand the mechanisms of Alzheimer's disease, but research is complicated by aggregate inhomogeneity and instability. We previously engineered a hairpin form of Aβ called Aβcc, which forms stable protofibrils that do not convert into amyloid fibrils. Here we provide a detailed characterization of Aβ42cc protofibrils. Like wild type Aβ they appear as smooth rod-like particles with a diameter of 3.1 (±0.2) nm and typical lengths in the range 60 to 220 nm when observed by atomic force microscopy. Non-perturbing analytical ultracentrifugation and nanoparticle tracking analyses are consistent with such rod-like protofibrils. Aβ42cc protofibrils bind the ANS dye indicating that they, like other toxic protein aggregates, expose hydrophobic surface. Assays with the OC/A11 pair of oligomer specific antibodies put Aβ42cc protofibrils into the same class of species as fibrillar oligomers of wild type Aβ. Aβ42cc protofibrils may be used to extract binding proteins in biological fluids and apolipoprotein E is readily detected as a binder in human serum. Finally, Aβ42cc protofibrils act to attenuate spontaneous synaptic activity in mouse hippocampal neurons. The experiments indicate considerable structural and chemical similarities between protofibrils formed by Aβ42cc and aggregates of wild type Aβ42. We suggest that Aβ42cc protofibrils may be used in research and applications that require stable preparations of protofibrillar Aβ.
Conflict of interest statement
Figures


Similar articles
-
Binding of human proteins to amyloid-β protofibrils.ACS Chem Biol. 2015 Mar 20;10(3):766-74. doi: 10.1021/cb5008663. Epub 2014 Dec 12. ACS Chem Biol. 2015. PMID: 25469473
-
Protofibrillar and Fibrillar Amyloid-β Binding Proteins in Cerebrospinal Fluid.J Alzheimers Dis. 2018;66(3):1053-1064. doi: 10.3233/JAD-180596. J Alzheimers Dis. 2018. PMID: 30372682
-
Stabilization of neurotoxic Alzheimer amyloid-beta oligomers by protein engineering.Proc Natl Acad Sci U S A. 2010 Aug 31;107(35):15595-600. doi: 10.1073/pnas.1001740107. Epub 2010 Aug 16. Proc Natl Acad Sci U S A. 2010. PMID: 20713699 Free PMC article.
-
Protein engineering to stabilize soluble amyloid β-protein aggregates for structural and functional studies.FEBS J. 2011 Oct;278(20):3884-92. doi: 10.1111/j.1742-4658.2011.08295.x. Epub 2011 Sep 6. FEBS J. 2011. PMID: 21824290 Review.
-
Amyloid-beta aggregates formed at polar-nonpolar interfaces differ from amyloid-beta protofibrils produced in aqueous buffers.Microsc Res Tech. 2005 Jul;67(3-4):164-74. doi: 10.1002/jemt.20189. Microsc Res Tech. 2005. PMID: 16103999 Review.
Cited by
-
Evaluation of Nanoparticle Tracking for Characterization of Fibrillar Protein Aggregates.AIChE J. 2014 Apr 1;60(4):1236-1244. doi: 10.1002/aic.14349. AIChE J. 2014. PMID: 25843955 Free PMC article.
-
Identification of proteins that specifically recognize and bind protofibrillar aggregates of amyloid-β.Sci Rep. 2017 Jul 20;7(1):5949. doi: 10.1038/s41598-017-06377-8. Sci Rep. 2017. PMID: 28729665 Free PMC article.
-
Protein nanofibrils and their use as building blocks of sustainable materials.RSC Adv. 2021 Dec 8;11(62):39188-39215. doi: 10.1039/d1ra06878d. eCollection 2021 Dec 6. RSC Adv. 2021. PMID: 35492452 Free PMC article. Review.
-
Amyloid-beta 1-40 is associated with alterations in NG2+ pericyte population ex vivo and in vitro.Aging Cell. 2018 Jun;17(3):e12728. doi: 10.1111/acel.12728. Epub 2018 Feb 17. Aging Cell. 2018. PMID: 29453790 Free PMC article.
-
Molecular Simulation of the Binding of Amyloid Beta to Apolipoprotein A-I in High-Density Lipoproteins.Int J Mol Sci. 2025 Feb 6;26(3):1380. doi: 10.3390/ijms26031380. Int J Mol Sci. 2025. PMID: 39941148 Free PMC article.
References
-
- Querfurth HW, LaFerla FM (2010) Alzheimer's disease. N Engl J Med 362: 329–344. - PubMed
-
- Hepler RW, Grimm KM, Nahas DD, Breese R, Dodson EC, et al. (2006) Solution state characterization of amyloid β-derived diffusible ligands. Biochemistry 45: 15157–15167. - PubMed
-
- Lesné S, Koh MT, Kotilinek L, Kayed R, Glabe CG, et al. (2006) A specific amyloid-β protein assembly in the brain impairs memory. Nature 440: 352–357. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

