In silico investigation of the short QT syndrome, using human ventricle models incorporating electromechanical coupling
- PMID: 23847545
- PMCID: PMC3701879
- DOI: 10.3389/fphys.2013.00166
In silico investigation of the short QT syndrome, using human ventricle models incorporating electromechanical coupling
Abstract
Introduction: Genetic forms of the Short QT Syndrome (SQTS) arise due to cardiac ion channel mutations leading to accelerated ventricular repolarization, arrhythmias and sudden cardiac death. Results from experimental and simulation studies suggest that changes to refractoriness and tissue vulnerability produce a substrate favorable to re-entry. Potential electromechanical consequences of the SQTS are less well-understood. The aim of this study was to utilize electromechanically coupled human ventricle models to explore electromechanical consequences of the SQTS.
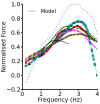
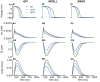
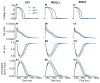
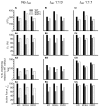
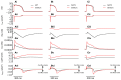
Methods and results: The Rice et al. mechanical model was coupled to the ten Tusscher et al. ventricular cell model. Previously validated K(+) channel formulations for SQT variants 1 and 3 were incorporated. Functional effects of the SQTS mutations on [Ca(2+)] i transients, sarcomere length shortening and contractile force at the single cell level were evaluated with and without the consideration of stretch-activated channel current (I sac). Without I sac, at a stimulation frequency of 1Hz, the SQTS mutations produced dramatic reductions in the amplitude of [Ca(2+)] i transients, sarcomere length shortening and contractile force. When I sac was incorporated, there was a considerable attenuation of the effects of SQTS-associated action potential shortening on Ca(2+) transients, sarcomere shortening and contractile force. Single cell models were then incorporated into 3D human ventricular tissue models. The timing of maximum deformation was delayed in the SQTS setting compared to control.
Conclusion: The incorporation of I sac appears to be an important consideration in modeling functional effects of SQT 1 and 3 mutations on cardiac electro-mechanical coupling. Whilst there is little evidence of profoundly impaired cardiac contractile function in SQTS patients, our 3D simulations correlate qualitatively with reported evidence for dissociation between ventricular repolarization and the end of mechanical systole.
Keywords: 3D model; human ventricles; mechanical contraction; short QT syndrome; stretch-activated channel.
Figures





References
-
- Alvarez B. V., Pérez N. G., Ennis I. L., Camilión de Hurtado M. C., Cingolani H. E. (1999). Mechanisms underlying the increase in force and Ca(2+) transient that follow stretch of cardiac muscle: a possible explanation of the Anrep effect. Circ. Res. 85, 716–722 10.1161/01.RES.85.8.716 - DOI - PubMed
-
- Antzelevitch C., Pollevick G. D., Cordeiro J. M., Casis O., Sanguinetti M. C., Aizawa Y., et al. (2007). Loss-of-function mutations in the cardiac calcium channel underlie a new clinical entity characterized by ST-segment elevation, short QT intervals, and sudden cardiac death. Circulation 115, 442–449 10.1161/CIRCULATIONAHA.106.668392 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

