Transition-state inhibitors of purine salvage and other prospective enzyme targets in malaria
- PMID: 23859211
- PMCID: PMC3819805
- DOI: 10.4155/fmc.13.51
Transition-state inhibitors of purine salvage and other prospective enzyme targets in malaria
Abstract
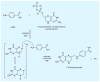
Malaria is a leading cause of human death within the tropics. The gradual generation of drug resistance imposes an urgent need for the development of new and selective antimalarial agents. Kinetic isotope effects coupled to computational chemistry have provided the relevant details on geometry and charge of enzymatic transition states to facilitate the design of transition-state analogs. These features have been reproduced into chemically stable mimics through synthetic chemistry, generating inhibitors with dissociation constants in the pico- to femto-molar range. Transition-state analogs are expected to contribute to the control of malaria.
Figures

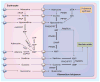

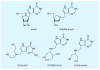






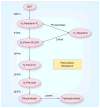
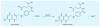


References
-
- Svenson JE, MacLean JD, Gyorkos TW, Keystone J. Imported malaria. Clinical presentation and examination of symptomatic travelers. Arch Intern Med. 1995;155(8):861–868. - PubMed
-
- Holt RA, Subramanian GM, Halpern A, et al. The genome sequence of the malaria mosquito Anopheles gambiae. Science. 2002;298(5591):129–149. - PubMed
-
- White NJ, Breman JG. Malaria. In: Anthony SF, Dennis LK, Dan LL, et al., editors. Harrison’s Principles of Internal Medicine. The McGraw-Hill Companies; NY, USA: 2008. pp. 1280–1294.
-
- Foley M, Tilley L. Quinoline antimalarials: mechanisms of action and resistance and prospects for new agents. Pharmacol Ther. 1998;79(1):55–87. - PubMed
Websites
-
- Centers for Disease Control and Prevention. Malaria Biology. www.cdc.gov/malaria/about/biology.
-
- WHO. World Malaria Report. 2011 www.who.int/malaria/world_malaria_report_2011/en.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
