The redox proteome
- PMID: 23861437
- PMCID: PMC3772199
- DOI: 10.1074/jbc.R113.464131
The redox proteome
Abstract
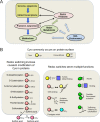
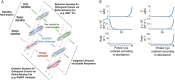
The redox proteome consists of reversible and irreversible covalent modifications that link redox metabolism to biologic structure and function. These modifications, especially of Cys, function at the molecular level in protein folding and maturation, catalytic activity, signaling, and macromolecular interactions and at the macroscopic level in control of secretion and cell shape. Interaction of the redox proteome with redox-active chemicals is central to macromolecular structure, regulation, and signaling during the life cycle and has a central role in the tolerance and adaptability to diet and environmental challenges.
Keywords: Glutathione; Redox Regulation; Systems Biology; Thiol; Thioredoxin.
Figures


References
-
- Keilin D. (1966) The History of Cell Respiration and Cytochrome, Cambridge University Press, Cambridge
-
- Mitchell P. (1979) Keilin's respiratory chain concept and its chemiosmotic consequences. Science 206, 1148–1159 - PubMed
-
- Mason H. S., Fowlks W. L., Peterson J. (1955) Oxygen transfer and electron transport by the phenolase complex. J. Am. Chem. Soc. 77, 2914–2915
-
- Hayaishi O., Katagiri M., Rothberg S. (1955) Mechanism of the pyrocatechase reaction. J. Am. Chem. Soc. 77, 5450–5451
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

