Acyl-CoA thioesterase 9 (ACOT9) in mouse may provide a novel link between fatty acid and amino acid metabolism in mitochondria
- PMID: 23864032
- PMCID: PMC11114068
- DOI: 10.1007/s00018-013-1422-1
Acyl-CoA thioesterase 9 (ACOT9) in mouse may provide a novel link between fatty acid and amino acid metabolism in mitochondria
Abstract
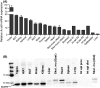
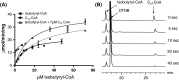
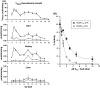
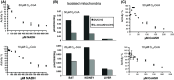
Acyl-CoA thioesterase (ACOT) activities are found in prokaryotes and in several compartments of eukaryotes where they hydrolyze a wide range of acyl-CoA substrates and thereby regulate intracellular acyl-CoA/CoA/fatty acid levels. ACOT9 is a mitochondrial ACOT with homologous genes found from bacteria to humans and in this study we have carried out an in-depth kinetic characterization of ACOT9 to determine its possible physiological function. ACOT9 showed unusual kinetic properties with activity peaks for short-, medium-, and saturated long-chain acyl-CoAs with highest V max with propionyl-CoA and (iso) butyryl-CoA while K cat/K m was highest with saturated long-chain acyl-CoAs. Further characterization of the short-chain acyl-CoA activity revealed that ACOT9 also hydrolyzes a number of short-chain acyl-CoAs and short-chain methyl-branched CoA esters that suggest a role for ACOT9 in regulation also of amino acid metabolism. In spite of markedly different K ms, ACOT9 can hydrolyze both short- and long-chain acyl-CoAs simultaneously, indicating that ACOT9 may provide a novel regulatory link between fatty acid and amino acid metabolism in mitochondria. Based on similar acyl-CoA chain-length specificities of recombinant ACOT9 and ACOT activity in mouse brown adipose tissue and kidney mitochondria, we conclude that ACOT9 is the major mitochondrial ACOT hydrolyzing saturated C2-C20-CoA in these tissues. Finally, ACOT9 activity is strongly regulated by NADH and CoA, suggesting that mitochondrial metabolic state regulates the function of ACOT9.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

