Clinical feasibility of noninvasive visualization of lymphatic flow with principles of spin labeling MR imaging: implications for lymphedema assessment
- PMID: 23864103
- PMCID: PMC4485559
- DOI: 10.1148/radiol.13120145
Clinical feasibility of noninvasive visualization of lymphatic flow with principles of spin labeling MR imaging: implications for lymphedema assessment
Abstract
Purpose: To extend a commonly used noninvasive arterial spin labeling magnetic resonance (MR) imaging method for measuring blood flow to evaluate lymphatic flow.
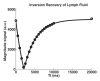
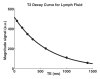
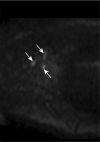
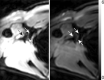
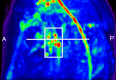
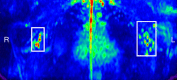
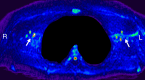
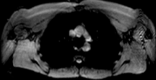
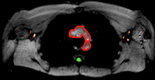
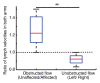
Materials and methods: All volunteers (n = 12) provided informed consent in accordance with institutional review board and HIPAA regulations. Quantitative relaxation time (T1 and T2) measurements were made in extracted human lymphatic fluid at 3.0 T. Guided by these parameters, an arterial spin labeling MR imaging approach was adapted to measure lymphatic flow (flow-alternating inversion-recovery lymphatic water labeling, 3 × 3 × 5 mm) in healthy subjects (n = 6; mean age, 30 years ± 1 [standard deviation]; recruitment duration, 2 months). Lymphatic flow velocity was quantified by performing spin labeling measurements as a function of postlabeling delay time and by measuring time to peak signal intensity in axillary lymph nodes. Clinical feasibility was evaluated in patients with stage II lymphedema (three women; age range, 43-64 years) and in control subjects with unilateral cuff-induced lymphatic stenosis (one woman, two men; age range, 31-35 years).
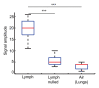
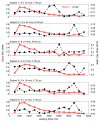
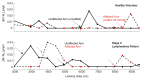
Results: Mean T1 and T2 relaxation times of lymphatic fluid at 3.0 T were 3100 msec ± 160 (range, 2930-3210 msec; median, 3200 msec) and 610 msec ± 12 (range, 598-618 msec; median, 610 msec), respectively. Healthy lymphatic flow (afferent vessel to axillary node) velocity was 0.61 cm/min ± 0.13 (n = 6). A reduction (P < .005) in lymphatic flow velocity in the affected arms of patients and the affected arms of healthy subjects with manipulated cuff-induced flow reduction was observed. The ratio of unaffected to affected axilla lymphatic velocity (1.24 ± 0.18) was significantly (P < .005) higher than the left-to-right ratio in healthy subjects (0.91 ± 0.18).
Conclusion: This work provides a foundation for clinical investigations whereby lymphedema etiogenesis and therapies may be interrogated without exogenous agents and with clinically available imaging equipment. Online supplemental material is available for this article.
© RSNA, 2013.
Figures
References
-
- Moseley AL, Carati CJ, Piller NB . A systematic review of common conservative therapies for arm lymphoedema secondary to breast cancer treatment . Ann Oncol 2007. ; 18 ( 4 ): 639 – 646 . - PubMed
-
- Armer J, Fu MR, Wainstock JM, Zagar E, Jacobs LK . Lymphedema following breast cancer treatment, including sentinel lymph node biopsy . Lymphology 2004. ; 37 ( 2 ): 73 – 91 . - PubMed
-
- Warren AG, Brorson H, Borud LJ, Slavin SA . Lymphedema: a comprehensive review . Ann Plast Surg 2007. ; 59 ( 4 ): 464 – 472 . - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

