CXCR7 is induced by hypoxia and mediates glioma cell migration towards SDF-1α
- PMID: 23865743
- PMCID: PMC3728118
- DOI: 10.1186/1471-2407-13-347
CXCR7 is induced by hypoxia and mediates glioma cell migration towards SDF-1α
Abstract
Background: Glioblastomas, the most common and malignant brain tumors of the central nervous system, exhibit high invasive capacity, which hinders effective therapy. Therefore, intense efforts aimed at improved therapeutics are ongoing to delineate the molecular mechanisms governing glioma cell migration and invasion.
Methods: In order to perform the studies, we employed optimal cell culture methods and hypoxic conditions, lentivirus-mediated knockdown of protein expression, Western Blot analysis, migration assays and immunoprecipitation. We determined statistical significance by unpaired t-test.
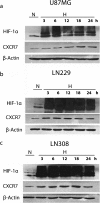
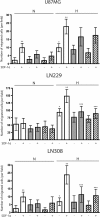
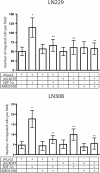
Results: In this report, we show that U87MG, LN229 and LN308 glioma cells express CXCR7 and that exposure to hypoxia upregulates CXCR7 protein expression in these cell lines. CXCR7-expressing U87MG, LN229 and LN308 glioma cells migrated towards stromal-derived factor (SDF)-1α/CXCL12 in hypoxic conditions in the Boyden chamber assays. While shRNA-mediated knockdown of CXCR7 expression did not affect the migration of any of the three cell lines in normoxic conditions, we observed a reduction in the migration of LN229 and LN308, but not U87MG, glioma cells towards SDF-1α in hypoxic conditions. In addition, knockdown of CXCR7 expression in LN229 and LN308 glioma cells decreased levels of SDF-1α-induced phosphorylation of ERK1/2 and Akt. Inhibiting CXCR4 in LN229 and LN308 glioma cells that were knocked down for CXCR7 did not further reduce migration towards SDF-1α in hypoxic conditions and did not affect the levels of phosphorylated ERK1/2 and Akt. Analysis of immunoprecipitated CXCR4 from LN229 and LN308 glioma cells revealed co-precipitated CXCR7.
Conclusions: Taken together, our findings indicate that both CXCR4 and CXCR7 mediate glioma cell migration towards SDF-1α in hypoxic conditions and support the development of therapeutic agents targeting these receptors.
Figures



Similar articles
-
HGF upregulates CXCR4 expression in gliomas via NF-kappaB: implications for glioma cell migration.J Neurooncol. 2010 Aug;99(1):33-40. doi: 10.1007/s11060-010-0111-2. Epub 2010 Feb 16. J Neurooncol. 2010. PMID: 20157762 Free PMC article.
-
Overlapping and distinct role of CXCR7-SDF-1/ITAC and CXCR4-SDF-1 axes in regulating metastatic behavior of human rhabdomyosarcomas.Int J Cancer. 2010 Dec 1;127(11):2554-68. doi: 10.1002/ijc.25245. Int J Cancer. 2010. PMID: 20162608 Free PMC article.
-
The SDF-1/CXCR4 axis is involved in adipose-derived stem cell migration.Neurourol Urodyn. 2024 Nov;43(8):2279-2289. doi: 10.1002/nau.25571. Epub 2024 Aug 16. Neurourol Urodyn. 2024. PMID: 39149821
-
The peculiarities of the SDF-1/CXCL12 system: in some cells, CXCR4 and CXCR7 sing solos, in others, they sing duets.Cell Tissue Res. 2014 Feb;355(2):239-53. doi: 10.1007/s00441-013-1747-y. Epub 2013 Nov 29. Cell Tissue Res. 2014. PMID: 24292718 Review.
-
Role of chemokine receptors CXCR4 and CXCR7 for platelet function.Biochem Soc Trans. 2015 Aug;43(4):720-6. doi: 10.1042/BST20150113. Epub 2015 Aug 3. Biochem Soc Trans. 2015. PMID: 26551719 Review.
Cited by
-
Aggressive migration in acidic pH of a glioblastoma cancer stem cell line in vitro is independent of ASIC and KCa3.1 ion channels, but involves phosphoinositide 3-kinase.Pflugers Arch. 2023 Mar;475(3):405-416. doi: 10.1007/s00424-022-02781-w. Epub 2022 Dec 16. Pflugers Arch. 2023. PMID: 36522586 Free PMC article.
-
Elevated CXCL12 in the plasma membrane of locally advanced rectal cancer after neoadjuvant chemoradiotherapy: a potential prognostic marker.J Cancer. 2022 Jan 1;13(1):162-173. doi: 10.7150/jca.64082. eCollection 2022. J Cancer. 2022. PMID: 34976180 Free PMC article.
-
Heterogeneous expression of the atypical chemokine receptor ACKR3 in glioblastoma patient-derived tissue samples and cell cultures.Sci Rep. 2024 Sep 20;14(1):21925. doi: 10.1038/s41598-024-73064-w. Sci Rep. 2024. PMID: 39300240 Free PMC article.
-
Transfection with GLS2 Glutaminase (GAB) Sensitizes Human Glioblastoma Cell Lines to Oxidative Stress by a Common Mechanism Involving Suppression of the PI3K/AKT Pathway.Cancers (Basel). 2019 Jan 19;11(1):115. doi: 10.3390/cancers11010115. Cancers (Basel). 2019. PMID: 30669455 Free PMC article.
-
Chemotactic responses of neural stem cells to SDF-1α correlate closely with their differentiation status.J Mol Neurosci. 2014;54(2):219-33. doi: 10.1007/s12031-014-0279-6. Epub 2014 Mar 22. J Mol Neurosci. 2014. PMID: 24659235
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

