Myomaker is a membrane activator of myoblast fusion and muscle formation
- PMID: 23868259
- PMCID: PMC3739301
- DOI: 10.1038/nature12343
Myomaker is a membrane activator of myoblast fusion and muscle formation
Abstract
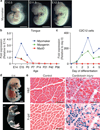
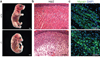
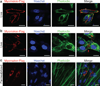
Fusion of myoblasts is essential for the formation of multi-nucleated muscle fibres. However, the identity of muscle-specific proteins that directly govern this fusion process in mammals has remained elusive. Here we identify a muscle-specific membrane protein, named myomaker, that controls myoblast fusion. Myomaker is expressed on the cell surface of myoblasts during fusion and is downregulated thereafter. Overexpression of myomaker in myoblasts markedly enhances fusion, and genetic disruption of myomaker in mice causes perinatal death due to an absence of multi-nucleated muscle fibres. Remarkably, forced expression of myomaker in fibroblasts promotes fusion with myoblasts, demonstrating the direct participation of this protein in the fusion process. Pharmacological perturbation of the actin cytoskeleton abolishes the activity of myomaker, consistent with previous studies implicating actin dynamics in myoblast fusion. These findings reveal a long-sought myogenic fusion protein that controls mammalian myoblast fusion and provide new insights into the molecular underpinnings of muscle formation.
Figures


References
-
- Chen EH, Olson EN. Unveiling the mechanisms of cell-cell fusion. Science. 2005;308:369–373. - PubMed
-
- Berkes CA, Tapscott SJ. MyoD and the transcriptional control of myogenesis. Semin Cell Dev Biol. 2005;16:585–595. - PubMed
-
- Buckingham M. Myogenic progenitor cells and skeletal myogenesis in vertebrates. Curr Opin Genet Dev. 2006;16:525–532. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

