Pb remobilization by bacterially mediated dissolution of pyromorphite Pb5(PO4)3Cl in presence of phosphate-solubilizing Pseudomonas putida
- PMID: 23872890
- PMCID: PMC3880484
- DOI: 10.1007/s11356-013-1968-3
Pb remobilization by bacterially mediated dissolution of pyromorphite Pb5(PO4)3Cl in presence of phosphate-solubilizing Pseudomonas putida
Abstract
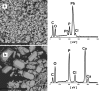
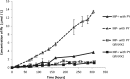
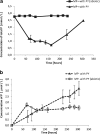
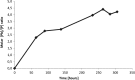
Remediation of lead (Pb)-contaminated sites with phosphate amendments is one of the best studied and cost-effective methods for in situ immobilization. In this treatment, a very stable mineral, pyromorphite Pb5(PO4)3Cl, is formed. Several studies propose to improve this treatment method with the addition of phosphate-solubilizing bacteria (PSB). The effect of bacteria on solubilization of pyromorphite is unknown. In this study, the effect of the soil microorganisms on the stability of pyromorphite Pb5(PO4)3Cl has been investigated in a set of batch solution experiments. The mineral was reacted with Pseudomonas putida, a common soil microorganism. Dissolution of pyromorphite was enhanced by the presence of P. putida, resulting in an elevated Pb concentration in the solution. This occurred even when the bacteria were provided with an additional source of phosphate in the solution. Pyromorphite has been shown to be a potential source of nutrient phosphorus for common soil bacteria. Thus, the use of PSB in remediation treatments of Pb contaminated sites may have adverse long-term impacts on Pb immobilization. Conscious phosphate management is suggested for long-term sustainability of the in situ Pb immobilization by pyromorphite formation.
Figures


Similar articles
-
Solubilization of Pb-bearing apatite Pb5(PO4)3Cl by bacteria isolated from polluted environment.Chemosphere. 2017 Mar;171:302-307. doi: 10.1016/j.chemosphere.2016.12.056. Epub 2016 Dec 21. Chemosphere. 2017. PMID: 28027474
-
Efficient lead immobilization by bio-beads containing Pseudomonas rhodesiae and bone char.J Hazard Mater. 2023 Apr 5;447:130772. doi: 10.1016/j.jhazmat.2023.130772. Epub 2023 Jan 13. J Hazard Mater. 2023. PMID: 36680905
-
Transforming cerussite to pyromorphite by immobilising Pb(II) using hydroxyapatite and Pseudomonas rhodesiae.Chemosphere. 2022 Jan;287(Pt 2):132235. doi: 10.1016/j.chemosphere.2021.132235. Epub 2021 Sep 11. Chemosphere. 2022. PMID: 34826926
-
Phosphate application to firing range soils for Pb immobilization: the unclear role of phosphate.J Hazard Mater. 2007 Jun 1;144(1-2):1-14. doi: 10.1016/j.jhazmat.2007.02.008. Epub 2007 Feb 12. J Hazard Mater. 2007. PMID: 17360110 Review.
-
Amending soils with phosphate as means to mitigate soil lead hazard: a critical review of the state of the science.J Toxicol Environ Health B Crit Rev. 2013;16(6):337-80. doi: 10.1080/10937404.2013.825216. J Toxicol Environ Health B Crit Rev. 2013. PMID: 24151967 Review.
Cited by
-
Long-Term in Situ Reduction in Soil Lead Bioavailability Measured in a Mouse Model.Environ Sci Technol. 2018 Dec 4;52(23):13908-13913. doi: 10.1021/acs.est.8b04684. Epub 2018 Nov 14. Environ Sci Technol. 2018. PMID: 30358995 Free PMC article.
-
Healthier Communities of Phytoplankton and Bacteria Achieved via the Application of Modified Clay in Shrimp Aquaculture Ponds.Int J Environ Res Public Health. 2021 Nov 4;18(21):11569. doi: 10.3390/ijerph182111569. Int J Environ Res Public Health. 2021. PMID: 34770083 Free PMC article.
-
Metal leakage from orthodontic appliances chemically alters enamel surface during experimental in vitro simulated treatment.Sci Rep. 2024 Mar 5;14(1):5412. doi: 10.1038/s41598-024-56111-4. Sci Rep. 2024. PMID: 38443566 Free PMC article.
-
Dietary Lead and Phosphate Interactions Affect Oral Bioavailability of Soil Lead in the Mouse.Environ Sci Technol. 2019 Nov 5;53(21):12556-12564. doi: 10.1021/acs.est.9b02803. Epub 2019 Oct 16. Environ Sci Technol. 2019. PMID: 31557437 Free PMC article.
References
-
- Bajda T. Solubility of mimetite Pb5(AsO4)3Cl at 5–55 °C. Environ Chem. 2010;7:268–278. doi: 10.1071/EN10021. - DOI
-
- Baker WE. An X-ray diffraction study of synthetic members of the pyromorphite series. Am Mineral. 1966;51:1712–1721.
-
- Bardgett RD (2006) The biology of soil: a community and ecosystem approach. The biology of habitats series. Oxford University Press, Oxford
-
- Chang JS, Law R, Chang CC. Biosorption of lead, copper and cadmium by biomass of Pseudomonas aeruginosa PU21. Water Res. 1997;31:1651–1658. doi: 10.1016/S0043-1354(97)00008-0. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

