The distribution of Dishevelled in convergently extending mesoderm
- PMID: 23876427
- PMCID: PMC3793869
- DOI: 10.1016/j.ydbio.2013.07.012
The distribution of Dishevelled in convergently extending mesoderm
Abstract
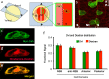
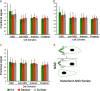
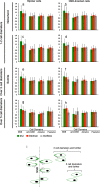
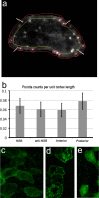
Convergent extension (CE) is a conserved morphogenetic movement that drives axial lengthening of the primary body axis and depends on the planar cell polarity (PCP) pathway. In Drosophila epithelia, a polarised subcellular accumulation of PCP core components, such as Dishevelled (Dvl) protein, is associated with PCP function. Dvl has long been thought to accumulate in the mediolateral protrusions in Xenopus chordamesoderm cells undergoing CE. Here we present a quantitative analysis of Dvl intracellular localisation in Xenopus chordamesoderm cells. We find that, surprisingly, accumulations previously observed at mediolateral protrusions of chordamesodermal cells are not protrusion-specific but reflect yolk-free cytoplasm and are quantitatively matched by the distribution of the cytoplasm-filling lineage marker dextran. However, separating cell cortex-associated from bulk Dvl signal reveals a statistical enrichment of Dvl in notochord-somite boundary-(NSB)-directed protrusions, which is dependent upon NSB proximity. Dvl puncta were also observed, but only upon elevated overexpression. These puncta showed no statistically significant spatial bias, in contrast to the strongly posteriorly-enriched GFP-Dvl puncta previously reported in zebrafish. We propose that Dvl distribution is more subtle and dynamic than previously appreciated and that in vertebrate mesoderm it reflects processes other than protrusion as such.
Keywords: Convergent extension; Dishevelled; Image analysis; Localisation; Planar cell polarity; QuimP; Xenopus.
© 2013 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
NEDD4L regulates convergent extension movements in Xenopus embryos via Disheveled-mediated non-canonical Wnt signaling.Dev Biol. 2014 Aug 1;392(1):15-25. doi: 10.1016/j.ydbio.2014.05.003. Epub 2014 May 14. Dev Biol. 2014. PMID: 24833518
-
Bimodal regulation of Dishevelled function by Vangl2 during morphogenesis.Hum Mol Genet. 2017 Jun 1;26(11):2053-2061. doi: 10.1093/hmg/ddx095. Hum Mol Genet. 2017. PMID: 28334810 Free PMC article.
-
FGF3 in the floor plate directs notochord convergent extension in the Ciona tadpole.Development. 2009 Jan;136(1):23-8. doi: 10.1242/dev.029157. Epub 2008 Nov 26. Development. 2009. PMID: 19036800 Free PMC article.
-
Dishevelled: The hub of Wnt signaling.Cell Signal. 2010 May;22(5):717-27. doi: 10.1016/j.cellsig.2009.11.021. Epub 2009 Dec 13. Cell Signal. 2010. PMID: 20006983 Review.
-
Dishevelled: in vivo roles of a multifunctional gene family during development.Curr Top Dev Biol. 2012;101:213-35. doi: 10.1016/B978-0-12-394592-1.00007-7. Curr Top Dev Biol. 2012. PMID: 23140631 Review.
Cited by
-
Cell cortex regulation by the planar cell polarity protein Prickle1.J Cell Biol. 2022 Jul 4;221(7):e202008116. doi: 10.1083/jcb.202008116. Epub 2022 May 5. J Cell Biol. 2022. PMID: 35512799 Free PMC article.
-
A dynamic intracellular distribution of Vangl2 accompanies cell polarization during zebrafish gastrulation.Development. 2015 Jul 15;142(14):2508-20. doi: 10.1242/dev.119032. Epub 2015 Jun 10. Development. 2015. PMID: 26062934 Free PMC article.
-
PAPC mediates self/non-self-distinction during Snail1-dependent tissue separation.J Cell Biol. 2015 Mar 16;208(6):839-56. doi: 10.1083/jcb.201409026. J Cell Biol. 2015. PMID: 25778923 Free PMC article.
-
Planar cell polarity in moving cells: think globally, act locally.Development. 2017 Jan 15;144(2):187-200. doi: 10.1242/dev.122804. Development. 2017. PMID: 28096212 Free PMC article. Review.
-
Spatial and temporal analysis of PCP protein dynamics during neural tube closure.Elife. 2018 Aug 6;7:e36456. doi: 10.7554/eLife.36456. Elife. 2018. PMID: 30080139 Free PMC article.
References
-
- Aigouy B., Farhadifar R., Staple D.B., Sagner A., Roper J.C., Julicher F., Eaton S. Cell flow reorients the axis of planar polarity in the wing epithelium of Drosophila. Cell. 2010;142:773–786. - PubMed
-
- Bosgraaf L., van Haastert P.J., Bretschneider T. Analysis of cell movement by simultaneous quantification of local membrane displacement and fluorescent intensities using Quimp2. Cell Motil. Cytoskeleton. 2009;66:156–165. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

