Modulation of biotransformation systems and ABC transporters by benznidazole in rats
- PMID: 23877690
- PMCID: PMC3811452
- DOI: 10.1128/AAC.02531-12
Modulation of biotransformation systems and ABC transporters by benznidazole in rats
Abstract
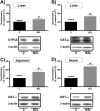
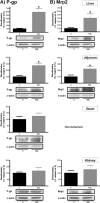
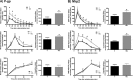
The effect of antichagasic benznidazole (BZL; 100 mg/kg body weight/day, 3 consecutive days, intraperitoneally) on biotransformation systems and ABC transporters was evaluated in rats. Expression of cytochrome P-450 (CYP3A), UDP-glucuronosyltransferase (UGT1A), glutathione S-transferases (alpha glutathione S-transferase [GST-α], GST-μ, and GST-π), multidrug-resistance-associated protein 2 (Mrp2), and P glycoprotein (P-gp) in liver, small intestine, and kidney was estimated by Western blotting. Increases in hepatic CYP3A (30%) and GST-μ (40%) and in intestinal GST-α (72% in jejunum and 136% in ileum) were detected. Significant increases in Mrp2 (300%) and P-gp (500%) proteins in liver from BZL-treated rats were observed without changes in kidney. P-gp and Mrp2 were also increased by BZL in jejunum (170% and 120%, respectively). In ileum, only P-gp was increased by BZL (50%). The activities of GST, P-gp, and Mrp2 correlated well with the upregulation of proteins in liver and jejunum. Plasma decay of a test dose of BZL (5 mg/kg body weight) administered intraduodenally was faster (295%) and the area under the concentration-time curve (AUC) was lower (41%) for BZL-pretreated rats than for controls. The biliary excretion of BZL was higher (60%) in the BZL group, and urinary excretion of BZL did not show differences between groups. The amount of absorbed BZL in intestinal sacs was lower (25%) in pretreated rats than in controls. In conclusion, induction of biotransformation enzymes and/or transporters by BZL could increase the clearance and/or decrease the intestinal absorption of coadministered drugs that are substrates of these systems, including BZL itself.
Figures
Similar articles
-
Regulation of biotransformation systems and ABC transporters by benznidazole in HepG2 cells: involvement of pregnane X-receptor.PLoS Negl Trop Dis. 2012;6(12):e1951. doi: 10.1371/journal.pntd.0001951. Epub 2012 Dec 13. PLoS Negl Trop Dis. 2012. PMID: 23272261 Free PMC article.
-
Up-regulation of ATP-binding cassette transporters in the THP-1 human macrophage cell line by the antichagasic benznidazole.Mem Inst Oswaldo Cruz. 2016 Nov;111(11):707-711. doi: 10.1590/0074-02760160080. Epub 2016 Oct 24. Mem Inst Oswaldo Cruz. 2016. PMID: 27783718 Free PMC article.
-
Effect of glucagon-like peptide 2 on hepatic, renal, and intestinal disposition of 1-chloro-2,4-dinitrobenzene.Drug Metab Dispos. 2012 Jul;40(7):1252-8. doi: 10.1124/dmd.111.044339. Epub 2012 Mar 27. Drug Metab Dispos. 2012. PMID: 22453052 Free PMC article.
-
Modulation of expression/function of intestinal P-glycoprotein under disease states.Expert Opin Drug Metab Toxicol. 2020 Jan;16(1):59-78. doi: 10.1080/17425255.2020.1701653. Epub 2019 Dec 10. Expert Opin Drug Metab Toxicol. 2020. PMID: 31821048 Review.
-
The glutathione S-transferase supergene family: regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance.Crit Rev Biochem Mol Biol. 1995;30(6):445-600. doi: 10.3109/10409239509083491. Crit Rev Biochem Mol Biol. 1995. PMID: 8770536 Review.
Cited by
-
Benznidazole Extended-Release Tablets for Improved Treatment of Chagas Disease: Preclinical Pharmacokinetic Study.Antimicrob Agents Chemother. 2016 Mar 25;60(4):2492-8. doi: 10.1128/AAC.02506-15. Print 2016 Apr. Antimicrob Agents Chemother. 2016. PMID: 26883698 Free PMC article.
-
Analysis of Time-Dependent Pharmacokinetics Using In Vitro-In Vivo Extrapolation and Physiologically Based Pharmacokinetic Modeling.Pharmaceutics. 2022 Nov 22;14(12):2562. doi: 10.3390/pharmaceutics14122562. Pharmaceutics. 2022. PMID: 36559055 Free PMC article.
-
Differential tissue distribution of discrete typing units after drug combination therapy in experimental Trypanosoma cruzi mixed infection.Parasitology. 2021 Nov;148(13):1595-1601. doi: 10.1017/S0031182021001281. Epub 2021 Jul 21. Parasitology. 2021. PMID: 35060468 Free PMC article.
-
Pharmacokinetics of Benznidazole in Experimental Chronic Chagas Disease Using the Swiss Mouse-Berenice-78 Trypanosoma cruzi Strain Model.Antimicrob Agents Chemother. 2021 Jan 20;65(2):e01383-20. doi: 10.1128/AAC.01383-20. Print 2021 Jan 20. Antimicrob Agents Chemother. 2021. PMID: 33168611 Free PMC article.
-
Pharmacokinetics and Tissue Distribution of Benznidazole after Oral Administration in Mice.Antimicrob Agents Chemother. 2017 Mar 24;61(4):e02410-16. doi: 10.1128/AAC.02410-16. Print 2017 Apr. Antimicrob Agents Chemother. 2017. PMID: 28167558 Free PMC article. Clinical Trial.
References
-
- World Health Organization 2007. Global plan to combat neglected tropical diseases 2008–2015 World Health Organization, Geneva, Switzerland
-
- Rassi A, Jr, Rassi A, Marín-Neto JA. 2010. Chagas disease. Lancet 375:1388–1402 - PubMed
-
- Gascon J, Bern C, Pinazzo MJ. 2010. Chagas disease in Spain, the United States and other non-endemic countries. Acta Trop. 115:22–27 - PubMed
-
- Maya JD, Cassels BK, Iturriaga-Vásquez P, Ferreira J, Faundez M, Galanti N, Ferreira A, Morello A. 2007. Mode of action of natural and synthetic drugs against Trypanosoma cruzi and their interaction with the mammalian host. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 146:601–620 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

