The MDM2-p53 pathway revisited
- PMID: 23885265
- PMCID: PMC3721034
- DOI: 10.7555/JBR.27.20130030
The MDM2-p53 pathway revisited
Abstract
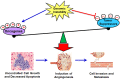
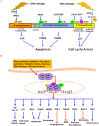
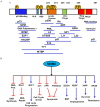
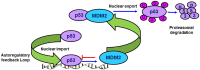
The p53 tumor suppressor is a key transcription factor regulating cellular pathways such as DNA repair, cell cycle, apoptosis, angiogenesis, and senescence. It acts as an important defense mechanism against cancer onset and progression, and is negatively regulated by interaction with the oncoprotein MDM2. In human cancers, the TP53 gene is frequently mutated or deleted, or the wild-type p53 function is inhibited by high levels of MDM2, leading to downregulation of tumor suppressive p53 pathways. Thus, the inhibition of MDM2-p53 interaction presents an appealing therapeutic strategy for the treatment of cancer. However, recent studies have revealed the MDM2-p53 interaction to be more complex involving multiple levels of regulation by numerous cellular proteins and epigenetic mechanisms, making it imperative to reexamine this intricate interplay from a holistic viewpoint. This review aims to highlight the multifaceted network of molecules regulating the MDM2-p53 axis to better understand the pathway and exploit it for anticancer therapy.
Keywords: MDM2-p53 interaction; cancer therapy; oncogene; tumor suppressor.
Conflict of interest statement
The authors reported no conflict of interests.
Figures
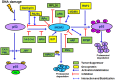
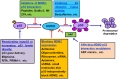
Similar articles
-
The role of tumor protein 53 mutations in common human cancers and targeting the murine double minute 2-p53 interaction for cancer therapy.Iran J Med Sci. 2012 Mar;37(1):3-8. Iran J Med Sci. 2012. PMID: 23115424 Free PMC article.
-
Efficient activation of p53 pathway in A549 cells exposed to L2, a novel compound targeting p53-MDM2 interaction.Anticancer Drugs. 2009 Jul;20(6):416-24. doi: 10.1097/cad.0b013e32832aa7b0. Anticancer Drugs. 2009. PMID: 19579266
-
MDM2 oncogene as a novel target for human cancer therapy.Curr Pharm Des. 2000 Mar;6(4):393-416. doi: 10.2174/1381612003400911. Curr Pharm Des. 2000. PMID: 10788589 Review.
-
P53-MDM2 Pathway: Evidences for A New Targeted Therapeutic Approach in B-Acute Lymphoblastic Leukemia.Front Pharmacol. 2016 Dec 16;7:491. doi: 10.3389/fphar.2016.00491. eCollection 2016. Front Pharmacol. 2016. PMID: 28018226 Free PMC article. Review.
-
Natural products targeting the p53-MDM2 pathway and mutant p53: Recent advances and implications in cancer medicine.Genes Dis. 2018 Jul 20;5(3):204-219. doi: 10.1016/j.gendis.2018.07.002. eCollection 2018 Sep. Genes Dis. 2018. PMID: 30320185 Free PMC article. Review.
Cited by
-
MDMX in Cancer: A Partner of p53 and a p53-Independent Effector.Biologics. 2024 Jan 31;18:61-78. doi: 10.2147/BTT.S436629. eCollection 2024. Biologics. 2024. PMID: 38318098 Free PMC article. Review.
-
Systems biology analysis reveals role of MDM2 in diabetic nephropathy.JCI Insight. 2016 Oct 20;1(17):e87877. doi: 10.1172/jci.insight.87877. JCI Insight. 2016. PMID: 27777973 Free PMC article.
-
Prognostic and therapeutic value of the Hippo pathway, RABL6A, and p53-MDM2 axes in sarcomas.Oncotarget. 2021 Apr 13;12(8):740-755. doi: 10.18632/oncotarget.27928. eCollection 2021 Apr 13. Oncotarget. 2021. PMID: 33889298 Free PMC article.
-
Wiring miRNAs to pathways: a topological approach to integrate miRNA and mRNA expression profiles.Nucleic Acids Res. 2014 Jun;42(11):e96. doi: 10.1093/nar/gku354. Epub 2014 May 6. Nucleic Acids Res. 2014. PMID: 24803669 Free PMC article.
-
The Roles of MDM2 and MDMX in Cancer.Annu Rev Pathol. 2016 May 23;11:617-44. doi: 10.1146/annurev-pathol-012414-040349. Epub 2016 Mar 17. Annu Rev Pathol. 2016. PMID: 27022975 Free PMC article. Review.
References
-
- Croce CM. Oncogenes and cancer. N Engl J Med. 2008;358:502–11. - PubMed
-
- Zhang Z, Li M, Rayburn ER, Hill DL, Zhang R, Wang H. Oncogenes as novel targets for cancer therapy (part II): Intermediate signaling molecules. Am J Pharmacogenomics. 2005;5:247–57. - PubMed
-
- Zhang Z, Li M, Rayburn ER, Hill DL, Zhang R, Wang H. Oncogenes as novel targets for cancer therapy (part III): transcription factors. Am J Pharmacogenomics. 2005;5:327–38. - PubMed
-
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74. - PubMed
-
- Perera S, Bapat B. Genetic Instability in Cancer. Atlas Genet CytogenetOncolHaematol. 2007 Jan; URL: http://AtlasGeneticsOncology.org/Deep/GenetInstabilityCancerID20056.html.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

