Neutral sphingomyelinase 2 modulates cytotoxic effects of protopanaxadiol on different human cancer cells
- PMID: 23889969
- PMCID: PMC3729373
- DOI: 10.1186/1472-6882-13-194
Neutral sphingomyelinase 2 modulates cytotoxic effects of protopanaxadiol on different human cancer cells
Abstract
Background: Some of ginsenosides, root extracts from Panax ginseng, exert cytotoxicity against cancer cells through disruption of membrane subdomains called lipid rafts. Protopanaxadiol (PPD) exhibits the highest cytotoxic effect among 8 ginsenosides which we evaluated for anti-cancer activity. We investigated if PPD disrupts lipid rafts in its cytotoxic effects and also the possible mechanisms.
Methods: Eight ginsenosides were evaluated using different cancer cells and cell viability assays. The potent ginsenoside, PPD was investigated for its roles in lipid raft disruption and downstream pathways to apoptosis of cancer cells. Anti-cancer effects of PPD was also investigated in vivo using mouse xenograft model.
Results: PPD consistently exerts its potent cytotoxicity in 2 cell survival assays using 5 different cancer cell lines. PPD disrupts lipid rafts in different ways from methyl-β-cyclodextrin (MβCD) depleting cholesterol out of the subdomains, since lipid raft proteins were differentially modulated by the saponin. During disruption of lipid rafts, PPD activated neutral sphingomyelinase 2 (nSMase 2) hydrolyzing membrane sphingomyelins into pro-apoptotic intracellular ceramides. Furthermore, PPD demonstrated its anti-cancer activities against K562 tumor cells in mouse xenograft model, confirming its potential as an adjunct or chemotherapeutic agent by itself in vivo.
Conclusions: This study demonstrates that neutral sphingomyelinase 2 is responsible for the cytotoxicity of PPD through production of apoptotic ceramides from membrane sphingomyelins. Thus neutral sphingomyelinase 2 and its relevant mechanisms may potentially be employed in cancer chemotherapies.
Figures
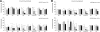
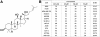

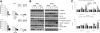

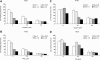
References
-
- Babina IS, Donatello S, Nabi IR, Hopkins AM. In: Breast cancer - carcinogenesis, cell growth and signalling pathways. Gunduz M, Gunduz E, editor. New York: INTECH; 2011. Lipid Rafts as Master Regulators of Breast Cancer Cell Function; pp. 401–428.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

