OnabotulinumtoxinA improves quality of life in patients with neurogenic detrusor overactivity
- PMID: 23892704
- PMCID: PMC3908462
- DOI: 10.1212/WNL.0b013e3182a2ca4d
OnabotulinumtoxinA improves quality of life in patients with neurogenic detrusor overactivity
Abstract
Objective: To evaluate the effects of onabotulinumtoxinA on patient-reported outcomes including health-related quality of life (HRQOL), treatment satisfaction, and treatment goal attainment in patients with urinary incontinence (UI) due to neurogenic detrusor overactivity (NDO).
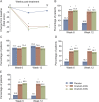
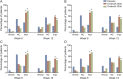
Methods: In this multicenter, double-blind, randomized, placebo-controlled, phase III, 52-week study (ClinicalTrials.gov NCT00311376), patients with UI due to NDO who were not adequately managed with anticholinergic therapy were treated with intradetrusor injections of onabotulinumtoxinA (200 or 300 U) or placebo (0.9% saline). HRQOL measures included the Incontinence Quality of Life (I-QOL) Questionnaire total score, and the 3 domain scores (avoidance and limiting behavior, psychosocial, and social embarrassment), the modified Overactive Bladder Patient Satisfaction with Treatment Questionnaire (OAB-PSTQ), and Patient Global Assessment. Assessments were made at baseline, posttreatment week 6 (primary time point), week 12, and at 12-week intervals.
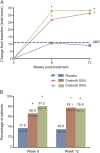
Results: Patients (mean age of 46 years with 30.5 weekly UI episodes at baseline) were randomized to receive placebo (n = 149) or onabotulinumtoxinA (200 U [n = 135] or 300 U [n = 132]). At week 6, improvements from baseline in I-QOL Questionnaire total score were greater (p < 0.001) in both onabotulinumtoxinA-treated groups vs placebo. Responses to the OAB-PSTQ also demonstrated greater mean improvements from baseline (p < 0.001) in both onabotulinumtoxinA-treated groups vs placebo at week 6. Patients who received onabotulinumtoxinA also reported greater improvement in the Patient Global Assessment than those in the placebo group (p ≤ 0.001 vs placebo).
Conclusions: Patients with UI due to NDO reported greater improvement in HRQOL and treatment satisfaction with onabotulinumtoxinA than with placebo consistently across several patient-reported outcome instruments.
Classification of evidence: This study provides Class I evidence that onabotulinumtoxinA intradetrusor injections (200 or 300 U) can improve quality of life measures in patients with NDO not adequately managed with anticholinergic therapy.
Figures
Similar articles
-
Treatment satisfaction and improvement in health-related quality of life with onabotulinumtoxinA in patients with urinary incontinence due to neurogenic detrusor overactivity.Neurourol Urodyn. 2013 Mar;32(3):242-9. doi: 10.1002/nau.22293. Epub 2012 Sep 10. Neurourol Urodyn. 2013. PMID: 22965657 Clinical Trial.
-
Efficacy and safety of onabotulinumtoxinA in patients with urinary incontinence due to neurogenic detrusor overactivity: a randomised, double-blind, placebo-controlled trial.Eur Urol. 2011 Oct;60(4):742-50. doi: 10.1016/j.eururo.2011.07.002. Epub 2011 Jul 13. Eur Urol. 2011. PMID: 21798658 Clinical Trial.
-
OnabotulinumtoxinA 100 U significantly improves all idiopathic overactive bladder symptoms and quality of life in patients with overactive bladder and urinary incontinence: a randomised, double-blind, placebo-controlled trial.Eur Urol. 2013 Aug;64(2):249-56. doi: 10.1016/j.eururo.2013.04.001. Epub 2013 Apr 10. Eur Urol. 2013. PMID: 23608668 Clinical Trial.
-
Efficacy of abobotulinumtoxinA versus onabotulinumtoxinA for the treatment of refractory neurogenic detrusor overactivity: a systematic review and indirect treatment comparison.J Med Econ. 2023 Jan-Dec;26(1):200-207. doi: 10.1080/13696998.2023.2165366. J Med Econ. 2023. PMID: 36647624
-
Chapter 5: Clinical data in neurogenic detrusor overactivity (NDO) and overactive bladder (OAB).Neurourol Urodyn. 2014 Jul;33 Suppl 3:S26-31. doi: 10.1002/nau.22630. Neurourol Urodyn. 2014. PMID: 25042140 Review.
Cited by
-
Treatment of lower urinary tract symptoms in multiple sclerosis patients: Review of the literature and current guidelines.Can Urol Assoc J. 2017 Mar-Apr;11(3-4):E110-E115. doi: 10.5489/cuaj.4059. Epub 2017 Mar 16. Can Urol Assoc J. 2017. PMID: 28360957 Free PMC article. Review.
-
Treatment of neurogenic detrusor overactivity and overactive bladder with Botox (onabotulinumtoxinA): Development, insights, and impact.Medicine (Baltimore). 2023 Jul 1;102(S1):e32377. doi: 10.1097/MD.0000000000032377. Medicine (Baltimore). 2023. PMID: 37499088 Free PMC article. Clinical Trial.
-
The evaluation and management of refractory neurogenic overactive bladder.Curr Urol Rep. 2014 Oct;15(10):444. doi: 10.1007/s11934-014-0444-z. Curr Urol Rep. 2014. PMID: 25118853 Review.
-
Efficacy and Safety of OnabotulinumtoxinA in Patients with Neurogenic Detrusor Overactivity: A Systematic Review and Meta-Analysis of Randomized Controlled Trials.PLoS One. 2016 Jul 27;11(7):e0159307. doi: 10.1371/journal.pone.0159307. eCollection 2016. PLoS One. 2016. PMID: 27463810 Free PMC article.
-
Botulinum Toxin Use in Neurourology.Rev Urol. 2018;20(2):84-93. doi: 10.3903/riu0792. Rev Urol. 2018. PMID: 30288145 Free PMC article.
References
-
- Hollingworth W, Campbell JD, Kowalski J, et al. Exploring the impact of changes in neurogenic urinary incontinence frequency and condition-specific quality of life on preference-based outcomes. Qual Life Res 2010;19:323–331 - PubMed
-
- Schurch B, Denys P, Kozma CM, Reese PR, Slaton T, Barron RL. Botulinum toxin A improves the quality of life of patients with neurogenic urinary incontinence. Eur Urol 2007;52:850–858 - PubMed
-
- Schurch B, Denys P, Kozma CM, Reese PR, Slaton T, Barron R. Reliability and validity of the Incontinence Quality of Life questionnaire in patients with neurogenic urinary incontinence. Arch Phys Med Rehabil 2007;88:646–652 - PubMed
-
- Stohrer M, Blok B, Castro-Diaz D, et al. EAU guidelines on neurogenic lower urinary tract dysfunction. Eur Urol 2009;56:81–88 - PubMed
-
- Abrams P, Andersson KE. Muscarinic receptor antagonists for overactive bladder. BJU Int 2007;100:987–1006 - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
