Glycolipid transfer protein expression is affected by glycosphingolipid synthesis
- PMID: 23894633
- PMCID: PMC3722133
- DOI: 10.1371/journal.pone.0070283
Glycolipid transfer protein expression is affected by glycosphingolipid synthesis
Erratum in
-
Correction: Glycolipid Transfer Protein Expression Is Affected by Glycosphingolipid Synthesis.PLoS One. 2013 Oct 16;8(10):10.1371/annotation/712bb339-6073-4e62-9f68-b285caedd913. doi: 10.1371/annotation/712bb339-6073-4e62-9f68-b285caedd913. eCollection 2013. PLoS One. 2013. PMID: 29220844 Free PMC article.
Abstract
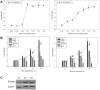
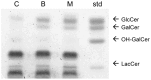
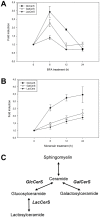
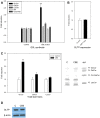
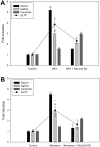
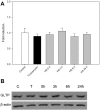
Members of the glycolipid transfer protein superfamily (GLTP) are found from animals and fungi to plants and red micro-alga. Eukaryotes that encode the glucosylceramide synthase responsible for the synthesis of glucosylceramide, the precursor for most glycosphingolipids, also produce GLTPs. Cells that does not synthesize glucosylceramide neither express GLTPs. Based on this genetic relationship there must be a strong correlation between the synthesis of glucosylceramide and GLTPs. To regulate the levels of glycolipids we have used inhibitors of intracellular trafficking, glycosphingolipid synthesis and degradation, and small interfering RNA to down-regulate the activity of glucosylceramide synthase activity. We found that GLTP expression, both at the mRNA and protein levels, is elevated in cells that accumulate glucosylceramide. Monensin and brefeldin A block intracellular vesicular transport mechanisms. Brefeldin A treatment leads to accumulation of newly synthesized glucosylceramide, galactosylceramide and lactosylceramide in a fused endoplasmic reticulum-Golgi complex. On the other hand, inhibiting glycosphingolipid degradation with conduritol-B-epoxide, that generates glucosylceramide accumulation in the lysosomes, did not affect the levels of GLTP. However, glycosphingolipid synthesis inhibitors like PDMP, NB-DNJ and myriocin, all decreased glucosylceramide and GLTP below normal levels. We also found that an 80% loss of glucosylceramide due to glucosylceramide synthase knockdown resulted in a significant reduction in the expression of GLTP. We show here that interfering with membrane trafficking events and simple neutral glycosphingolipid synthesis will affect the expression of GLTP. We postulate that a change in the glucosylceramide balance causes a response in the GLTP expression, and put forward that GLTP might play a role in lipid directing and sensing of glucosylceramide at the ER-Golgi interface.
Conflict of interest statement
Figures
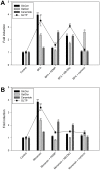
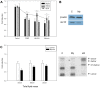
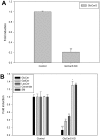
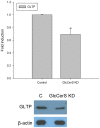
References
-
- Degroote S, Wolthoorn J, van Meer G (2004) The cell biology of glycosphingolipids. Semin Cell Dev Biol 15: 375–387. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

