Therapeutic hypothermia decreases phenytoin elimination in children with traumatic brain injury
- PMID: 23896831
- PMCID: PMC3783553
- DOI: 10.1097/CCM.0b013e318292316c
Therapeutic hypothermia decreases phenytoin elimination in children with traumatic brain injury
Abstract
Objective: Preclinical and clinical studies have suggested that therapeutic hypothermia, while decreasing neurologic injury, may also lead to drug toxicity that may limit its benefit. Cooling decreases cytochrome P450 (CYP)-mediated drug metabolism, and limited clinical data suggest that drug levels are elevated. Fosphenytoin is metabolized by cytochrome P450 2C, has a narrow therapeutic range, and is a commonly used antiepileptic medication. The objective of this study was to evaluate the impact of therapeutic hypothermia on phenytoin levels and pharmacokinetics in children with severe traumatic brain injury.
Design: Pharmacokinetic analysis of subjects participating in a multicenter randomized phase III study of therapeutic hypothermia for severe traumatic brain injury.
Setting: ICU at the Children's Hospital of Pittsburgh.
Patients: Nineteen children with severe traumatic brain injury.
Interventions: None.
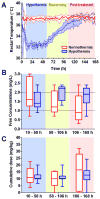
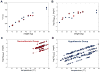
Measurements and main results: A sum of 121 total and 114 free phenytoin levels were evaluated retrospectively in 10 hypothermia-treated and nine normothermia-treated children who were randomized to 48 hours of cooling to 32-33°C followed by slow rewarming or controlled normothermia. Drug dosing, body temperatures, and demographics were collected during cooling, rewarming, and posttreatment periods (8 d). A trend toward elevated free phenytoin levels in the hypothermia group (p=0.051) to a median of 2.2 mg/L during rewarming was observed and was not explained by dosing differences. Nonlinear mixed-effects modeling incorporating both free and total levels demonstrated that therapeutic hypothermia specifically decreased the time-variant component of the maximum velocity of phenytoin metabolism (Vmax) 4.6-fold (11.6-2.53 mg/hr) and reduced the overall Vmax by ~50%. Simulations showed that the increased risk for drug toxicity extends many days beyond the end of the cooling period.
Conclusions: Therapeutic hypothermia significantly reduces phenytoin elimination in children with severe traumatic brain injury leading to increased drug levels for an extended period of time after cooling. Pharmacokinetic interactions between hypothermia and medications should be considered when caring for children receiving this therapy.
Conflict of interest statement
The rest of the authors have not disclosed any potential conflicts of interest.
Figures

Comment in
-
High level alert! Modeling temperature and phenytoin: appropriate risk management or virtual reality?Crit Care Med. 2013 Oct;41(10):2454-5. doi: 10.1097/CCM.0b013e31829641ca. Crit Care Med. 2013. PMID: 24060782 No abstract available.
References
-
- Bernard SA, Gray TW, Buist MD, et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346(8):557–563. - PubMed
-
- Hypothermia after Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med. 2002;346(8):549–556. - PubMed
-
- Azzopardi DV, Strohm B, Edwards AD, et al. Moderate hypothermia to treat perinatal asphyxial encephalopathy. N Engl J Med. 2009;361(14):1349–1358. - PubMed
-
- Eicher DJ, Wagner CL, Katikaneni LP, et al. Moderate hypothermia in neonatal encephalopathy: efficacy outcomes. Pediatr Neurol. 2005;32(1):11–17. - PubMed
-
- Gluckman PD, Wyatt JS, Azzopardi D, et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet. 2005;365(9460):663–670. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

