CD8+ T cells specific for a malaria cytoplasmic antigen form clusters around infected hepatocytes and are protective at the liver stage of infection
- PMID: 23897612
- PMCID: PMC3811763
- DOI: 10.1128/IAI.00570-13
CD8+ T cells specific for a malaria cytoplasmic antigen form clusters around infected hepatocytes and are protective at the liver stage of infection
Abstract
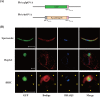
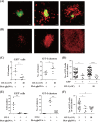
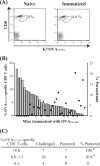
Following Anopheles mosquito-mediated introduction into a human host, Plasmodium parasites infect hepatocytes and undergo intensive replication. Accumulating evidence indicates that CD8(+) T cells induced by immunization with attenuated Plasmodium sporozoites can confer sterile immunity at the liver stage of infection; however, the mechanisms underlying this protection are not clearly understood. To address this, we generated recombinant Plasmodium berghei ANKA expressing a fusion protein of an ovalbumin epitope and green fluorescent protein in the cytoplasm of the parasite. We have shown that the ovalbumin epitope is presented by infected liver cells in a manner dependent on a transporter associated with antigen processing and becomes a target of specific CD8(+) T cells from the T cell receptor transgenic mouse line OT-I, leading to protection at the liver stage of Plasmodium infection. We visualized the interaction between OT-I cells and infected hepatocytes by intravital imaging using two-photon microscopy. OT-I cells formed clusters around infected hepatocytes, leading to the elimination of the intrahepatic parasites and subsequent formation of large clusters of OT-I cells in the liver. Gamma interferon expressed in CD8(+) T cells was dispensable for this protective response. Additionally, we found that polyclonal ovalbumin-specific memory CD8(+) T cells induced by de novo immunization were able to confer sterile protection, although the threshold frequency of the protection was relatively high. These studies revealed a novel mechanism of specific CD8(+) T cell-mediated protective immunity and demonstrated that proteins expressed in the cytoplasm of Plasmodium parasites can become targets of specific CD8(+) T cells during liver-stage infection.
Figures



References
-
- Nussenzweig RS, Vanderberg J, Most H, Orton C. 1967. Protective immunity produced by the injection of X-irradiated sporozoites of Plasmodium berghei. Nature 216:160–162 - PubMed
-
- Hoffman SL, Doolan DL. 2000. Malaria vaccines-targeting infected hepatocytes. Nat. Med. 6:1218–1219 - PubMed
-
- Mueller AK, Labaied M, Kappe SH, Matuschewski K. 2005. Genetically modified Plasmodium parasites as a protective experimental malaria vaccine. Nature 433:164–167 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

