Soluble ecto-5'-nucleotidase (5'-NT), alkaline phosphatase, and adenosine deaminase (ADA1) activities in neonatal blood favor elevated extracellular adenosine
- PMID: 23897810
- PMCID: PMC3779727
- DOI: 10.1074/jbc.M113.484212
Soluble ecto-5'-nucleotidase (5'-NT), alkaline phosphatase, and adenosine deaminase (ADA1) activities in neonatal blood favor elevated extracellular adenosine
Abstract
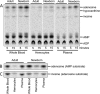
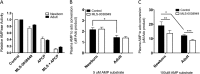
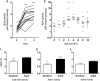
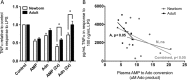
Extracellular adenosine, a key regulator of physiology and immune cell function that is found at elevated levels in neonatal blood, is generated by phosphohydrolysis of adenine nucleotides released from cells and catabolized by deamination to inosine. Generation of adenosine monophosphate (AMP) in blood is driven by cell-associated enzymes, whereas conversion of AMP to adenosine is largely mediated by soluble enzymes. The identities of the enzymes responsible for these activities in whole blood of neonates have been defined in this study and contrasted to adult blood. We demonstrate that soluble 5'-nucleotidase (5'-NT) and alkaline phosphatase (AP) mediate conversion of AMP to adenosine, whereas soluble adenosine deaminase (ADA) catabolizes adenosine to inosine. Newborn blood plasma demonstrates substantially higher adenosine-generating 5'-NT and AP activity and lower adenosine-metabolizing ADA activity than adult plasma. In addition to a role in soluble purine metabolism, abundant AP expressed on the surface of circulating neonatal neutrophils is the dominant AMPase on these cells. Plasma samples from infant observational cohorts reveal a relative plasma ADA deficiency at birth, followed by a gradual maturation of plasma ADA through infancy. The robust adenosine-generating capacity of neonates appears functionally relevant because supplementation with AMP inhibited whereas selective pharmacologic inhibition of 5'-NT enhanced Toll-like receptor-mediated TNF-α production in neonatal whole blood. Overall, we have characterized previously unrecognized age-dependent expression patterns of plasma purine-metabolizing enzymes that result in elevated plasma concentrations of anti-inflammatory adenosine in newborns. Targeted manipulation of purine-metabolizing enzymes may benefit this vulnerable population.
Keywords: ADP; AMP; ATP; Adenosine; Adenosine Receptor; Immunology; Infectious Diseases; Innate Immunity; Purine; Purinergic Agonists.
Figures



Similar articles
-
Serum adenosine deaminase, 5'-nucleotidase & alkaline phosphatase in breast cancer patients.Indian J Med Res. 1995 Jun;101:247-9. Indian J Med Res. 1995. PMID: 7672835 Clinical Trial.
-
Adenosine metabolism during phorbol myristate acetate-mediated induction of HL-60 cell differentiation: changes in expression pattern of adenosine kinase, adenosine deaminase, and 5'-nucleotidase.J Immunol. 1997 May 15;158(10):4947-52. J Immunol. 1997. PMID: 9144513
-
The ecto-enzymes CD73 and adenosine deaminase modulate 5'-AMP-derived adenosine in myofibroblasts of the rat small intestine.Purinergic Signal. 2018 Dec;14(4):409-421. doi: 10.1007/s11302-018-9623-6. Epub 2018 Sep 29. Purinergic Signal. 2018. PMID: 30269308 Free PMC article.
-
Enzymes involved in purine metabolism--a review of histochemical localization and functional implications.Histol Histopathol. 1999 Oct;14(4):1321-40. doi: 10.14670/HH-14.1321. Histol Histopathol. 1999. PMID: 10506947 Review.
-
Enzymes involved in metabolism of extracellular nucleotides and nucleosides: functional implications and measurement of activities.Crit Rev Biochem Mol Biol. 2014 Nov-Dec;49(6):473-97. doi: 10.3109/10409238.2014.953627. Crit Rev Biochem Mol Biol. 2014. PMID: 25418535 Review.
Cited by
-
Group B streptococci infection model shows decreased regulatory capacity of cord blood cells.Pediatr Res. 2022 Nov;92(5):1407-1416. doi: 10.1038/s41390-021-01880-1. Epub 2022 Feb 14. Pediatr Res. 2022. PMID: 35165359 Free PMC article.
-
Decreased circulating CD73 and adenosine deaminase are associated with disease severity in hospitalized patients with COVID-19.Int J Immunopathol Pharmacol. 2023 Jan-Dec;37:3946320231185703. doi: 10.1177/03946320231185703. Int J Immunopathol Pharmacol. 2023. PMID: 37364162 Free PMC article.
-
Vascular Calcification Revisited: A New Perspective for Phosphate Transport.Curr Cardiol Rev. 2015 Nov 6;11(4):341-351. doi: 10.2174/1573403X11666150805120505. Curr Cardiol Rev. 2015. PMID: 26242187 Free PMC article.
-
Targeting immunosuppressive adenosine in cancer.Nat Rev Cancer. 2017 Dec;17(12):709-724. doi: 10.1038/nrc.2017.86. Epub 2017 Oct 23. Nat Rev Cancer. 2017. PMID: 29059149 Review.
-
Structural and Functional Integration of Tissue-Nonspecific Alkaline Phosphatase Within the Alkaline Phosphatase Superfamily: Evolutionary Insights and Functional Implications.Metabolites. 2024 Nov 25;14(12):659. doi: 10.3390/metabo14120659. Metabolites. 2024. PMID: 39728440 Free PMC article. Review.
References
-
- Schenk U., Westendorf A. M., Radaelli E., Casati A., Ferro M., Fumagalli M., Verderio C., Buer J., Scanziani E., Grassi F. (2008) Purinergic control of T cell activation by ATP released through pannexin-1 hemichannels. Sci. Signal. 1, ra6. - PubMed
-
- Baricordi O. R., Ferrari D., Melchiorri L., Chiozzi P., Hanau S., Chiari E., Rubini M., Di Virgilio F. (1996) An ATP-activated channel is involved in mitogenic stimulation of human T lymphocytes. Blood 87, 682–690 - PubMed
-
- Baricordi O. R., Melchiorri L., Adinolfi E., Falzoni S., Chiozzi P., Buell G., Di Virgilio F. (1999) Increased proliferation rate of lymphoid cells transfected with the P2X7 ATP receptor. J. Biol. Chem. 274, 33206–33208 - PubMed
-
- Sud'ina G. F., Mirzoeva O. K., Galkina S. I., Pushkareva M. A., Ullrich V. (1998) Involvement of ecto-ATPase and extracellular ATP in polymorphonuclear granulocyte-endothelial interactions. FEBS Lett. 423, 243–248 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P01 HL087203/HL/NHLBI NIH HHS/United States
- R01 AR053102/AR/NIAMS NIH HHS/United States
- R01 HL062458/HL/NHLBI NIH HHS/United States
- T32 HD055148/HD/NICHD NIH HHS/United States
- R21 CA164970/CA/NCI NIH HHS/United States
- R01 HL094400/HL/NHLBI NIH HHS/United States
- MC_UP_A900_1122/MRC_/Medical Research Council/United Kingdom
- MC_UP_A900_1115/MRC_/Medical Research Council/United Kingdom
- N01 AI050023/AI/NIAID NIH HHS/United States
- T32 AI007061/AI/NIAID NIH HHS/United States
- R01 AI100135/AI/NIAID NIH HHS/United States
- R01 DE012889/DE/NIDCR NIH HHS/United States
- CAPMC/ CIHR/Canada
- 1R01AI100135-01/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

