Interaction of membrane/lipid rafts with the cytoskeleton: impact on signaling and function: membrane/lipid rafts, mediators of cytoskeletal arrangement and cell signaling
- PMID: 23899502
- PMCID: PMC3867519
- DOI: 10.1016/j.bbamem.2013.07.018
Interaction of membrane/lipid rafts with the cytoskeleton: impact on signaling and function: membrane/lipid rafts, mediators of cytoskeletal arrangement and cell signaling
Abstract
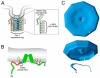
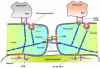
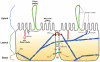
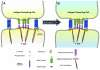
The plasma membrane in eukaryotic cells contains microdomains that are enriched in certain glycosphingolipids, gangliosides, and sterols (such as cholesterol) to form membrane/lipid rafts (MLR). These regions exist as caveolae, morphologically observable flask-like invaginations, or as a less easily detectable planar form. MLR are scaffolds for many molecular entities, including signaling receptors and ion channels that communicate extracellular stimuli to the intracellular milieu. Much evidence indicates that this organization and/or the clustering of MLR into more active signaling platforms depends upon interactions with and dynamic rearrangement of the cytoskeleton. Several cytoskeletal components and binding partners, as well as enzymes that regulate the cytoskeleton, localize to MLR and help regulate lateral diffusion of membrane proteins and lipids in response to extracellular events (e.g., receptor activation, shear stress, electrical conductance, and nutrient demand). MLR regulate cellular polarity, adherence to the extracellular matrix, signaling events (including ones that affect growth and migration), and are sites of cellular entry of certain pathogens, toxins and nanoparticles. The dynamic interaction between MLR and the underlying cytoskeleton thus regulates many facets of the function of eukaryotic cells and their adaptation to changing environments. Here, we review general features of MLR and caveolae and their role in several aspects of cellular function, including polarity of endothelial and epithelial cells, cell migration, mechanotransduction, lymphocyte activation, neuronal growth and signaling, and a variety of disease settings. This article is part of a Special Issue entitled: Reciprocal influences between cell cytoskeleton and membrane channels, receptors and transporters. Guest Editor: Jean Claude Hervé.
Keywords: AC; AD; AJ; AMPAR; APC; APP; Alzheimer's disease; Aβ; CAM; CBD; CRAC; CSD; CTX; Cav; Caveola; Caveolin; Cytoskeleton; EC; ECM; FAK; Flot; G-protein-coupled receptor; GD; GJ; GM; GPCR; GPI; ICAM/VCAM; Ion channel; JAM; MLR; MT; Membrane/lipid raft; N-methyl-D-aspartate receptor; NMDAR; PTRF; PrP; RTK; Signaling receptor; T cell receptor; TCR; TEM; TJ; TRPC1; TSPN; Trk; VGCC; adenylyl cyclases; adherent junctions; alpha-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate receptor; amyloid beta peptide; amyloid precursor protein; antigen presenting cell; cAMP; cSMAC; caveolin; caveolin binding domain; caveolin scaffolding domain; cellular adhesion molecules; central supramolecular activation cluster; cholera toxin; cholesterol recognition/interaction amino acid consensus; cyclic adenosine 3′,5′ monophosphate; eNOS; endothelial cell; endothelial nitric oxide synthase (NOS3); extracellular matrix; flotillin; focal adhesion kinase; ganglioside disialic acid; ganglioside monosialic acid; gap junctions; glycosylphosphatidylinositol; inter/vascular CAM; junctional adhesion molecules; mGluR; membrane/lipid rafts; metabotropic glutamate receptor; microtubules; pMHC; peripheral major histocompatibility complex; polymerase I and transcript release factor; prion protein; receptor tyrosine kinases; tetraspanin; tetraspanin-enriched microdomains; tight junctions; transient receptor potential cation channel; tropomyosin receptor kinase; voltage-gated Ca(2+) channels.
Published by Elsevier B.V.
Figures

References
-
- Mouritsen OG, Zuckermann MJ. What's so special about cholesterol? Lipids. 2004;39:1101–1113. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

