Hypothyroxinemia induced by mild iodine deficiency deregulats thyroid proteins during gestation and lactation in dams
- PMID: 23917811
- PMCID: PMC3774435
- DOI: 10.3390/ijerph10083233
Hypothyroxinemia induced by mild iodine deficiency deregulats thyroid proteins during gestation and lactation in dams
Abstract
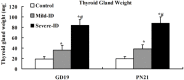
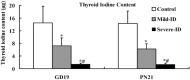
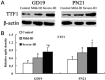
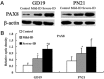
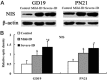
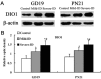
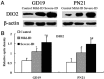
The main object of the present study was to explore the effect on thyroidal proteins following mild iodine deficiency (ID)-induced maternal hypothyroxinemia during pregnancy and lactation. In the present study, we established a maternal hypothyroxinemia model in female Wistar rats by using a mild ID diet. Maternal thyroid iodine content and thyroid weight were measured. Expressions of thyroid-associated proteins were analyzed. The results showed that the mild ID diet increased thyroid weight, decreased thyroid iodine content and increased expressions of thyroid transcription factor 1, paired box gene 8 and Na+/I- symporter on gestational day (GD) 19 and postpartum days (PN) 21 in the maternal thyroid. Moreover, the up-regulated expressions of type 1 iodothyronine deiodinase (DIO1) and type 2 iodothyronine deiodinase (DIO2) were detected in the mild ID group on GD19 and PN21. Taken together, our data indicates that during pregnancy and lactation, a maternal mild ID could induce hypothyroxinemia and increase the thyroidal DIO1 and DIO2 levels.
Figures
Similar articles
-
Prenatal iodine deficiency results in structurally and functionally immature lungs in neonatal rats.Am J Physiol Lung Cell Mol Physiol. 2012 May 15;302(10):L1037-43. doi: 10.1152/ajplung.00191.2011. Epub 2012 Mar 16. Am J Physiol Lung Cell Mol Physiol. 2012. PMID: 22427529
-
Maternal iodine supplementation improves motor coordination in offspring by modulating the mGluR1 signaling pathway in mild iodine deficiency-induced hypothyroxinemia rats.J Nutr Biochem. 2018 Aug;58:80-89. doi: 10.1016/j.jnutbio.2018.04.012. Epub 2018 May 1. J Nutr Biochem. 2018. PMID: 29886192
-
The expression of the sodium/iodide symporter is up-regulated in the thyroid of fetuses of iodine-deficient rats.Endocrinology. 2001 Sep;142(9):3736-41. doi: 10.1210/endo.142.9.8377. Endocrinology. 2001. PMID: 11517148
-
What happens to the normal thyroid during pregnancy?Thyroid. 1999 Jul;9(7):631-5. doi: 10.1089/thy.1999.9.631. Thyroid. 1999. PMID: 10447005 Review.
-
Iodine deficiency and brain development in the first half of pregnancy.Public Health Nutr. 2007 Dec;10(12A):1554-70. doi: 10.1017/S1368980007360928. Public Health Nutr. 2007. PMID: 18053280 Review.
Cited by
-
What Iodine Intervention Measures Should Be Taken in Different Water Iodine Areas? Evidence from a Cross-sectional Chinese Survey.Biol Trace Elem Res. 2022 Nov;200(11):4654-4663. doi: 10.1007/s12011-021-03050-0. Epub 2021 Dec 17. Biol Trace Elem Res. 2022. PMID: 34919209
-
Iodine excess exposure during pregnancy and lactation impairs maternal thyroid function in rats.Endocr Connect. 2017 Oct;6(7):510-521. doi: 10.1530/EC-17-0106. Epub 2017 Aug 16. Endocr Connect. 2017. PMID: 28814477 Free PMC article.
References
-
- De La Vieja A., Dohan O., Levy O., Carrasco N. Molecular analysis of the sodium iodide symporter impact on thyroid and extrathyroid pathophysiology. Physiol. Rev. 2000;80:1083–1105. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

