Procollagen III N-terminal propeptide and desmosine are released by matrix destruction in pulmonary tuberculosis
- PMID: 23922364
- PMCID: PMC3805234
- DOI: 10.1093/infdis/jit343
Procollagen III N-terminal propeptide and desmosine are released by matrix destruction in pulmonary tuberculosis
Abstract
Background: Tuberculosis is transmitted by patients with pulmonary disease. Matrix metalloproteinases (MMPs) drive lung destruction in tuberculosis but the resulting matrix degradation products (MDPs) have not been studied. We investigate the hypothesis that MMP activity generates matrix turnover products as correlates of lung pathology.
Methods: Induced sputum and plasma were collected prospectively from human immunodeficiency virus (HIV) positive and negative patients with pulmonary tuberculosis and controls. Concentrations of MDPs and MMPs were analyzed by ELISA and Luminex array in 2 patient cohorts.
Results: Procollagen III N-terminal propeptide (PIIINP) was 3.8-fold higher in induced sputum of HIV-uninfected tuberculosis patients compared to controls and desmosine, released during elastin degradation, was 2.4-fold higher. PIIINP was elevated in plasma of tuberculosis patients. Plasma PIIINP correlated with induced sputum MMP-1 concentrations and radiological scores, demonstrating that circulating MDPs reflect lung destruction. In a second patient cohort of mixed HIV seroprevalence, plasma PIIINP concentration was increased 3.0-fold above controls (P < .001). Plasma matrix metalloproteinase-8 concentrations were also higher in tuberculosis patients (P = .001). Receiver operating characteristic analysis utilizing these 2 variables demonstrated an area under the curve of 0.832 (P < .001).
Conclusions: In pulmonary tuberculosis, MMP-driven immunopathology generates matrix degradation products.
Keywords: extracellular matrix; immunopathology; lung; matrix metalloproteinase; mycobacteria.
Figures


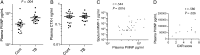

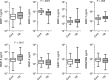

References
-
- Dye C, Williams BG. The population dynamics and control of tuberculosis. Science. 2010;328:856–61. - PubMed
-
- Frieden TR, Sterling TR, Munsiff SS, Watt CJ, Dye C. Tuberculosis. Lancet. 2003;362:887–99. - PubMed
-
- Walzl G, Ronacher K, Hanekom W, Scriba TJ, Zumla A. Immunological biomarkers of tuberculosis. Nat Rev Immunol. 2011;11:343–54. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 094000/WT_/Wellcome Trust/United Kingdom
- U2R TW007370/TW/FIC NIH HHS/United States
- U2R TW007370-01A1/TW/FIC NIH HHS/United States
- HHMI/Howard Hughes Medical Institute/United States
- U1175.02.002.00014.01/MRC_/Medical Research Council/United Kingdom
- 081667/WT_/Wellcome Trust/United Kingdom
- 088316/WT_/Wellcome Trust/United Kingdom
- MC_U117588499/MRC_/Medical Research Council/United Kingdom
- 084323/WT_/Wellcome Trust/United Kingdom
- DHCS/06/05/012/DH_/Department of Health/United Kingdom
- P30 AI060354/AI/NIAID NIH HHS/United States
- U2R TW007373/TW/FIC NIH HHS/United States
- U2R TW007373-01A1/TW/FIC NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

