Attenuation and immunogenicity of host-range extended modified vaccinia virus Ankara recombinants
- PMID: 23928462
- PMCID: PMC3787882
- DOI: 10.1016/j.vaccine.2013.07.057
Attenuation and immunogenicity of host-range extended modified vaccinia virus Ankara recombinants
Abstract
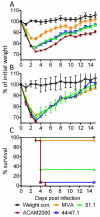
Modified vaccinia virus Ankara (MVA) is being widely investigated as a safe smallpox vaccine and as an expression vector to produce vaccines against other infectious diseases and cancer. MVA was isolated following more than 500 passages in chick embryo fibroblasts and suffered several major deletions and numerous small mutations resulting in replication defects in human and most other mammalian cells as well as severe attenuation of pathogenicity. Due to the host range restriction, primary chick embryo fibroblasts are routinely used for production of MVA-based vaccines. While a replication defect undoubtedly contributes to safety of MVA, it is worth considering whether host range and attenuation are partially separable properties. Marker rescue transfection experiments resulted in the creation of recombinant MVAs with extended mammalian cell host range. Here, we characterize two host-range extended rMVAs and show that they (i) have acquired the ability to stably replicate in Vero cells, which are frequently used as a cell substrate for vaccine manufacture, (ii) are severely attenuated in immunocompetent and immunodeficient mouse strains following intranasal infection, (iii) are more pathogenic than MVA but less pathogenic than the ACAM2000 vaccine strain at high intracranial doses, (iv) do not form lesions upon tail scratch in mice in contrast to ACAM2000 and (v) induce protective humoral and cell-mediated immune responses similar to MVA. The extended host range of rMVAs may be useful for vaccine production.
Keywords: Attenuated live vaccines; Recombinant vaccinia virus; Virus pathogenesis; Virus vectors.
Published by Elsevier Ltd.
Figures


Similar articles
-
SPI-1 is a missing host-range factor required for replication of the attenuated modified vaccinia Ankara (MVA) vaccine vector in human cells.PLoS Pathog. 2019 May 30;15(5):e1007710. doi: 10.1371/journal.ppat.1007710. eCollection 2019 May. PLoS Pathog. 2019. PMID: 31145755 Free PMC article.
-
Spontaneous and Targeted Mutations in the Decapping Enzyme Enhance Replication of Modified Vaccinia Virus Ankara (MVA) in Monkey Cells.J Virol. 2021 Sep 9;95(19):e0110421. doi: 10.1128/JVI.01104-21. Epub 2021 Sep 9. J Virol. 2021. PMID: 34232734 Free PMC article.
-
Introduction of the six major genomic deletions of modified vaccinia virus Ankara (MVA) into the parental vaccinia virus is not sufficient to reproduce an MVA-like phenotype in cell culture and in mice.J Virol. 2010 Oct;84(19):9907-19. doi: 10.1128/JVI.00756-10. Epub 2010 Jul 28. J Virol. 2010. PMID: 20668072 Free PMC article.
-
The Brighton Collaboration standardized template for collection of key information for risk/benefit assessment of a Modified Vaccinia Ankara (MVA) vaccine platform.Vaccine. 2021 May 21;39(22):3067-3080. doi: 10.1016/j.vaccine.2020.08.050. Epub 2020 Oct 17. Vaccine. 2021. PMID: 33077299 Free PMC article. Review.
-
Modified Vaccinia Virus Ankara: History, Value in Basic Research, and Current Perspectives for Vaccine Development.Adv Virus Res. 2017;97:187-243. doi: 10.1016/bs.aivir.2016.07.001. Epub 2016 Aug 1. Adv Virus Res. 2017. PMID: 28057259 Free PMC article. Review.
Cited by
-
Safety and immunogenicity of orally administered poxvirus vectored constructs in the white-footed mouse (Peromyscus leucopus).Vaccine X. 2022 Dec 28;13:100259. doi: 10.1016/j.jvacx.2022.100259. eCollection 2023 Apr. Vaccine X. 2022. PMID: 36654838 Free PMC article.
-
SPI-1 is a missing host-range factor required for replication of the attenuated modified vaccinia Ankara (MVA) vaccine vector in human cells.PLoS Pathog. 2019 May 30;15(5):e1007710. doi: 10.1371/journal.ppat.1007710. eCollection 2019 May. PLoS Pathog. 2019. PMID: 31145755 Free PMC article.
-
Generation of an Attenuated Tiantan Vaccinia Virus Strain by Deletion of Multiple Genes.Front Cell Infect Microbiol. 2017 Oct 31;7:462. doi: 10.3389/fcimb.2017.00462. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 29164070 Free PMC article.
-
Percutaneous Vaccination as an Effective Method of Delivery of MVA and MVA-Vectored Vaccines.PLoS One. 2016 Feb 19;11(2):e0149364. doi: 10.1371/journal.pone.0149364. eCollection 2016. PLoS One. 2016. PMID: 26895072 Free PMC article.
-
Identification of Vaccinia Virus Inhibitors and Cellular Functions Necessary for Efficient Viral Replication by Screening Bioactives and FDA-Approved Drugs.Vaccines (Basel). 2020 Jul 21;8(3):401. doi: 10.3390/vaccines8030401. Vaccines (Basel). 2020. PMID: 32708182 Free PMC article.
References
-
- Hochstein-Mintzel V, Huber HC, Stickl H. Virulenz und immunogenität eines modifizierten vaccinia-virus (Stamm MVA) [Virulence and immunogenicity of a modified vaccinia virus (strain MVA) Z Immun-Forsch. 1972;144:140–5. - PubMed
-
- Stickl H, Hochstein-Mintzel V, Mayr A, Huber HC, Schäfer H, Holzner A. MVA-stufenimpfung gegen pocken. klinische erprobung des attenuierten pocken-lebendimpfstoffes, stamm MVA (MVA vaccination against smallpox: clinical trials of an attenuated live vaccinia virus strain (MVA) Dtsch Med Wschr. 1974;99:2386–92. - PubMed
-
- Mayr A, Hochstein-Mintzel V, Stickl H. Abstammung, eigenschaften und verwendung des attenuierten vaccinia-stammes MVA (Passage history, properties, and applicability of the attenuated vaccinia virus strain MVA) Infection. 1975;3:6–14.
-
- Mayr A, Stickl H, Müller HK, Danner K, Singer H. Der pockenimpfstamm MVA: marker, genetische struktur, erfahrungen mit der parenteralen schutzimpfung und verhalten im abwehrgeschwächten organismus (The smallpox vaccination strain MVA: marker, genetic structure, experience gained with parenteral vaccination and behavior in organisms with debilitated defense mechanism. Zbl Bakt Hyg. 1978;167:375–90. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

