Aromatase in human liver and its diseases
- PMID: 23930207
- PMCID: PMC3699842
- DOI: 10.1002/cam4.85
Aromatase in human liver and its diseases
Abstract
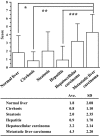
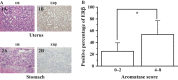
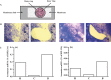
Estrogens play important roles in the cell proliferation and invasion of estrogen-dependent human neoplasms. Aromatase overexpression has been also reported in hepatitis and hepatocellular carcinoma (HCC) compared with normal liver but its details in these hepatic disorders have remained unclear. Therefore, in this study, we first immunolocalized aromatase using immunohistochemistry in patients with liver cirrhosis, steatosis, hepatitis, HCC, and metastasis liver carcinoma (MLC) in order to study the detailed status of intrahepatic aromatase. Aromatase immunoreactivity was predominantly detected in nonneoplastic hepatocytes around tumor cells. We then evaluated the effects of an interaction between hepatocytes and carcinoma cells upon aromatase mRNA expression, using HepG2 as a substitute model of hepatocytes by coculture systems. Aromatase mRNA levels in HepG2 were significantly increased by coculture with all carcinoma cell lines examined. We also evaluated alternative splicing of aromatase exon 1 but the same splicing variant was used in HepG2 cells regardless of carcinoma cell lines employed in the coculture system. These findings obtained in HepG2 indicated that carcinoma cells, whether metastatic or primary, induced aromatase expression in adjacent normal hepatocytes possibly through the soluble aromatase inducible factors in human hepatic microenvironments.
Keywords: Liver cancer; metastasis; pathology.
Figures



Similar articles
-
Merlin, the product of NF2 gene, is associated with aromatase expression and estrogen formation in human liver tissues and liver cancer cells.J Steroid Biochem Mol Biol. 2017 Sep;172:222-230. doi: 10.1016/j.jsbmb.2016.05.023. Epub 2016 Jun 8. J Steroid Biochem Mol Biol. 2017. PMID: 27289045 Review.
-
Expression of wild-type and variant estrogen receptor alpha in liver carcinogenesis and tumor progression.OMICS. 2011 May;15(5):313-7. doi: 10.1089/omi.2010.0108. Epub 2011 Feb 24. OMICS. 2011. PMID: 21348637
-
Intratumoral localization of aromatase and interaction between stromal and parenchymal cells in the non-small cell lung carcinoma microenvironment.Cancer Res. 2010 Aug 15;70(16):6659-69. doi: 10.1158/0008-5472.CAN-09-4653. Cancer Res. 2010. PMID: 20710045
-
Eukaryotic elongation factor-1α 2 knockdown inhibits hepatocarcinogenesis by suppressing PI3K/Akt/NF-κB signaling.World J Gastroenterol. 2016 Apr 28;22(16):4226-37. doi: 10.3748/wjg.v22.i16.4226. World J Gastroenterol. 2016. PMID: 27122673 Free PMC article.
-
Aromatase in nontumoral and malignant human liver tissues and cells.Ann N Y Acad Sci. 2009 Feb;1155:187-93. doi: 10.1111/j.1749-6632.2009.03706.x. Ann N Y Acad Sci. 2009. PMID: 19250204 Review.
Cited by
-
Sex Differences in the Regulation of Vasopressin and Oxytocin Secretion in Bile Duct-Ligated Rats.Neuroendocrinology. 2021;111(3):237-248. doi: 10.1159/000508104. Epub 2020 Apr 24. Neuroendocrinology. 2021. PMID: 32335554 Free PMC article.
-
Porcine intestinal lymphoid tissues synthesize estradiol.J Vet Sci. 2018 Jul 31;19(4):477-482. doi: 10.4142/jvs.2018.19.4.477. J Vet Sci. 2018. PMID: 29486537 Free PMC article.
-
The Influence of Sex Hormones in Liver Function and Disease.Cells. 2023 Jun 11;12(12):1604. doi: 10.3390/cells12121604. Cells. 2023. PMID: 37371074 Free PMC article. Review.
-
Imbalanced expression pattern of steroid receptor coactivator-1 and -3 in liver cancer compared with normal liver: An immunohistochemical study with tissue microarray.Oncol Lett. 2018 Nov;16(5):6339-6348. doi: 10.3892/ol.2018.9443. Epub 2018 Sep 17. Oncol Lett. 2018. PMID: 30405769 Free PMC article.
-
Add-On Selective Estrogen Receptor Modulators for Methadone Maintenance Treatment.Front Endocrinol (Lausanne). 2021 Aug 5;12:638884. doi: 10.3389/fendo.2021.638884. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34434167 Free PMC article.
References
-
- Harada N, Ota H, Yoshimura N, Katsuyama T, Takagi Y. Localized aberrant expression of cytochrome P450 aromatase in primary and metastatic malignant tumors of human liver. J. Clin. Endocrinol. Metab. 1998;83:697–702. - PubMed
-
- Nagasue N, Ito A, Yukaya H, Ogawa Y. Estrogen receptors in hepatocellular carcinoma. Cancer. 1986;57:87–91. - PubMed
-
- Villa E, Dugani A, Moles A, Camellini L, Grottola A, Buttafoco P, et al. Variant liver estrogen receptor transcripts already occur at an early stage of chronic liver disease. Hepatology. 1998;27:983–988. - PubMed
-
- Iavarone M, Lampertico P, Seletti C, Francesca Donato M, Ronchi G, et al. The clinical and pathogenetic significance of estrogen receptor-beta expression in chronic liver diseases and liver carcinoma. Cancer. 2003;98:529–534. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

