Kallikrein transduced mesenchymal stem cells protect against anti-GBM disease and lupus nephritis by ameliorating inflammation and oxidative stress
- PMID: 23935844
- PMCID: PMC3720854
- DOI: 10.1371/journal.pone.0067790
Kallikrein transduced mesenchymal stem cells protect against anti-GBM disease and lupus nephritis by ameliorating inflammation and oxidative stress
Abstract
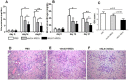
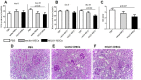
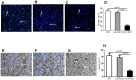
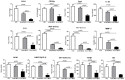
Previously we have shown that kallikreins (klks) play a renoprotective role in nephrotoxic serum induced nephritis. In this study, we have used mesenchymal stem cells (MSCs) as vehicles to deliver klks into the injured kidneys and have measured their therapeutic effect on experimental antibody induced nephritis and lupus nephritis. Human KLK-1 (hKLK1) gene was transduced into murine MSCs using a retroviral vector to generate a stable cell line, hKLK1-MSC, expressing high levels of hKLK1. 129/svj mice subjected to anti-GBM induced nephritis were transplanted with 10(6) hKLK1-MSCs and hKLK1 expression was confirmed in the kidneys. Compared with vector-MSCs injected mice, the hKLK1-MSCs treated mice showed significantly reduced proteinuria, blood urea nitrogen (BUN) and ameliorated renal pathology. Using the same strategy, we treated lupus-prone B6.Sle1.Sle3 bicongenic mice with hKLK1-MSCs and demonstrated that hKLK1-MSCs delivery also attenuated lupus nephritis. Mechanistically, hKLK1-MSCs reduced macrophage and T-lymphocyte infiltration into the kidney by suppressing the expression of inflammation cytokines. Moreover, hKLK1 transduced MSCs were more resistant to oxidative stress-induced apoptosis. These findings advance genetically modified MSCs as potential gene delivery tools for targeting therapeutic agents to the kidneys in order to modulate inflammation and oxidative stress in lupus nephritis.
Conflict of interest statement
Figures



Similar articles
-
Glutathione S-transferase Mu 2-transduced mesenchymal stem cells ameliorated anti-glomerular basement membrane antibody-induced glomerulonephritis by inhibiting oxidation and inflammation.Stem Cell Res Ther. 2014 Jan 30;5(1):19. doi: 10.1186/scrt408. Stem Cell Res Ther. 2014. PMID: 24480247 Free PMC article.
-
Delivering Oxidation Resistance-1 (OXR1) to Mouse Kidney by Genetic Modified Mesenchymal Stem Cells Exhibited Enhanced Protection against Nephrotoxic Serum Induced Renal Injury and Lupus Nephritis.J Stem Cell Res Ther. 2014 Sep 10;4(9):231. doi: 10.4172/2157-7633.1000231. J Stem Cell Res Ther. 2014. PMID: 25995969 Free PMC article.
-
Mesenchymal stem cells prevent podocyte injury in lupus-prone B6.MRL-Faslpr mice via polarizing macrophage into an anti-inflammatory phenotype.Nephrol Dial Transplant. 2019 Apr 1;34(4):597-605. doi: 10.1093/ndt/gfy195. Nephrol Dial Transplant. 2019. PMID: 29982691
-
Immunomodulatory Activity of Mesenchymal Stem Cells in Lupus Nephritis: Advances and Applications.Front Immunol. 2022 Mar 10;13:843192. doi: 10.3389/fimmu.2022.843192. eCollection 2022. Front Immunol. 2022. PMID: 35359961 Free PMC article. Review.
-
Experimental anti-GBM nephritis as an analytical tool for studying spontaneous lupus nephritis.Arch Immunol Ther Exp (Warsz). 2008 Jan-Feb;56(1):31-40. doi: 10.1007/s00005-008-0007-4. Epub 2008 Feb 5. Arch Immunol Ther Exp (Warsz). 2008. PMID: 18250969 Review.
Cited by
-
Glutathione S-transferase Mu 2-transduced mesenchymal stem cells ameliorated anti-glomerular basement membrane antibody-induced glomerulonephritis by inhibiting oxidation and inflammation.Stem Cell Res Ther. 2014 Jan 30;5(1):19. doi: 10.1186/scrt408. Stem Cell Res Ther. 2014. PMID: 24480247 Free PMC article.
-
How Stem and Progenitor Cells Can Affect Renal Diseases.Cells. 2024 Aug 30;13(17):1460. doi: 10.3390/cells13171460. Cells. 2024. PMID: 39273032 Free PMC article. Review.
-
A systems approach to renal inflammation in SLE.Clin Immunol. 2017 Dec;185:109-118. doi: 10.1016/j.clim.2016.08.015. Epub 2016 Aug 14. Clin Immunol. 2017. PMID: 27534926 Free PMC article. Review.
-
Evaluation of the Therapeutic Potential of Mesenchymal Stem Cells (MSCs) in Preclinical Models of Autoimmune Diseases.Stem Cells Int. 2022 Jul 28;2022:6379161. doi: 10.1155/2022/6379161. eCollection 2022. Stem Cells Int. 2022. PMID: 35935180 Free PMC article. Review.
-
Kallikrein 12 Regulates Innate Resistance of Murine Macrophages against Mycobacterium bovis Infection by Modulating Autophagy and Apoptosis.Cells. 2019 May 5;8(5):415. doi: 10.3390/cells8050415. Cells. 2019. PMID: 31060300 Free PMC article.
References
-
- Bomback AS, Appel GB (2010) Updates on the treatment of lupus nephritis. J Am Soc Nephrol 21: 2028–2035. - PubMed
-
- van der Vlag J, Berden JH (2011) Lupus nephritis: role of antinucleosome autoantibodies. Semin Nephrol 31: 376–389. - PubMed
-
- Zykova SN, Seredkina NE, Rekvig OP (2007) Glomerular targets for autoantibodies in lupus nephritis–an apoptotic origin. Ann N Y Acad Sci 1108: 1–10. - PubMed
-
- Contreras G, Pardo V, Leclercq B, Lenz O, Tozman E, et al. (2004) Sequential therapies for proliferative lupus nephritis. N Engl J Med 350: 971–980. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

