Suitability of Biomorphic Silicon Carbide Ceramics as Drug Delivery Systems against Bacterial Biofilms
- PMID: 23936680
- PMCID: PMC3725843
- DOI: 10.1155/2013/104529
Suitability of Biomorphic Silicon Carbide Ceramics as Drug Delivery Systems against Bacterial Biofilms
Abstract
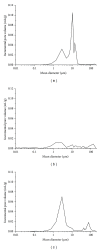
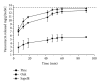
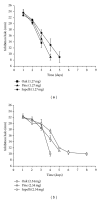
The present work is aimed at getting a new insight into biomorphic silicon carbides (bioSiCs) as bone replacement materials. BioSiCs from a variety of precursors were produced, characterized, and loaded with a broad-spectrum antibiotic. The capacity of loaded bioSiCs for preventing and/or treating preformed S. aureus biofilms has been studied. The differences in precursor characteristics are maintained after the ceramic production process. All bioSiCs allow the loading process by capillarity, giving loaded materials with drug release profiles dependent on their microstructure. The amount of antibiotic released in liquid medium during the first six hours depends on bioSiC porosity, but it could exceed the minimum inhibitory concentration of Staphylococcus aureus, for all the materials studied, thus preventing the proliferation of bacteria. Differences in the external surface and the number and size of open external pores of bioSiCs contribute towards the variations in the effect against bacteria when experiments are carried out using solid media. The internal structure and surface properties of all the systems seem to facilitate the therapeutic activity of the antibiotic on the preformed biofilms, reducing the number of viable bacteria present in the biofilm compared to controls.
Figures


References
-
- Davies DG, Parsek MR, Pearson JP, Iglewski BH, Costerton JW, Greenberg EP. The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science. 1998;280(5361):295–298. - PubMed
-
- Patel R. Biofilms and antimicrobial resistance. Clinical Orthopaedics and Related Research. 2005;437:41–47. - PubMed
-
- Costerton JW, Montanaro L, Arciola CR. Biofilm in implant infections: its production and regulation. International Journal of Artificial Organs. 2005;28(11):1062–1068. - PubMed
-
- Stoodley P, Sauer K, Davies DG, Costerton JW. Biofilms as complex differentiated communities. Annual Review of Microbiology. 2002;56:187–209. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

