Optimization of naltrexone diclofenac codrugs for sustained drug delivery across microneedle-treated skin
- PMID: 23943543
- PMCID: PMC3870048
- DOI: 10.1007/s11095-013-1147-8
Optimization of naltrexone diclofenac codrugs for sustained drug delivery across microneedle-treated skin
Abstract
Purpose: The purpose of this work was to optimize the structure of codrugs for extended delivery across microneedle treated skin. Naltrexone, the model compound was linked with diclofenac, a nonspecific cyclooxygenase inhibitor to enhance the pore lifetime following microneedle treatment and develop a 7 day transdermal system for naltrexone.
Methods: Four different codrugs of naltrexone and diclofenac were compared in terms of stability and solubility. Transdermal flux, permeability and skin concentration of both parent drugs and codrugs were quantified to form a structure permeability relationship.
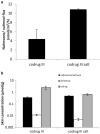
Results: The results indicated that all codrugs bioconverted in the skin. The degree of conversion was dependent on the structure, phenol linked codrugs were less stable compared to the secondary alcohol linked structures. The flux of naltrexone across microneedle treated skin and the skin concentration of diclofenac were higher for the phenol linked codrugs. The polyethylene glycol link enhanced solubility of the codrugs, which translated into flux enhancement.
Conclusion: The current studies indicated that formulation stability of codrugs and the flux of naltrexone can be enhanced via structure design optimization. The polyethylene glycol linked naltrexone diclofenac codrug is better suited for a 7 day drug delivery system both in terms of stability and drug delivery.
Figures




Similar articles
-
Development of a codrug approach for sustained drug delivery across microneedle-treated skin.J Pharm Sci. 2013 May;102(5):1458-67. doi: 10.1002/jps.23469. Epub 2013 Feb 15. J Pharm Sci. 2013. PMID: 23417751
-
In vitro permeation of a pegylated naltrexone prodrug across microneedle-treated skin.J Control Release. 2010 Aug 17;146(1):37-44. doi: 10.1016/j.jconrel.2010.05.034. Epub 2010 Jun 4. J Control Release. 2010. PMID: 20678989 Free PMC article.
-
Development of gellan gum containing formulations for transdermal drug delivery: Component evaluation and controlled drug release using temperature responsive nanogels.Int J Pharm. 2016 Jul 25;509(1-2):465-476. doi: 10.1016/j.ijpharm.2016.05.062. Epub 2016 Jun 1. Int J Pharm. 2016. PMID: 27260133
-
The codrug approach for facilitating drug delivery and bioactivity.Expert Opin Drug Deliv. 2016 Sep;13(9):1311-25. doi: 10.1080/17425247.2016.1187598. Epub 2016 May 27. Expert Opin Drug Deliv. 2016. PMID: 27159251 Review.
-
Enhanced transdermal drug delivery techniques: an extensive review of patents.Recent Pat Drug Deliv Formul. 2009 Jun;3(2):105-24. doi: 10.2174/187221109788452285. Recent Pat Drug Deliv Formul. 2009. PMID: 19519571 Review.
Cited by
-
Transdermal Drug Delivery Systems and Their Use in Obesity Treatment.Int J Mol Sci. 2021 Nov 25;22(23):12754. doi: 10.3390/ijms222312754. Int J Mol Sci. 2021. PMID: 34884558 Free PMC article. Review.
References
-
- Strasinger CL, Scheff NN, Stinchcomb AL. Prodrugs and codrugs as strategies for improving percutaneous absorption. Expert Rev Dermatol. 2008;3(2):221–33. 04/01; 2012/07.
-
- Stella V, Borchardt R, Hageman M, Oliyai R, Maag H, Tilley J. Prodrugs: challenges and rewards part 1. 1st ed. Springer; 2007.
-
- American Chemical Society https://scifinder.cas.org.
-
- McCaul ME, Wand GS, Rohde C, Lee SM. Serum 6-beta-naltrexol levels are related to alcohol responses in heavy drinkers. Alcohol Clin Exp Res. 2000;24(9):1385–91. - PubMed
-
- Verebey K, Volavka J, Mule SJ, Resnick RB. Naltrexone: disposition, metabolism, and effects after acute and chronic dosing. Clin Pharmacol Ther. 1976;20(3):315–28. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

