The safety of thoracentesis in patients with uncorrected bleeding risk
- PMID: 23952852
- PMCID: PMC3960907
- DOI: 10.1513/AnnalsATS.201210-088OC
The safety of thoracentesis in patients with uncorrected bleeding risk
Abstract
Background: Thoracentesis is commonly performed to evaluate pleural effusions. Many medications (warfarin, heparin, clopidogrel) or physiological factors (elevated International Normalized Ratio [INR], thrombocytopenia, uremia) increase the risk for bleeding. Frequently these medications are withheld or transfusions are performed to normalize physiological parameters before a procedure. The safety of performing thoracentesis without correction of these bleeding risks has not been prospectively evaluated.
Methods: This prospective observational cohort study enrolled 312 patients who underwent thoracentesis. All patients were evaluated for the presence of risk factors for bleeding. Hematocrit levels were obtained pre- and postprocedure, and the occurrence of postprocedural hemothorax was evaluated.
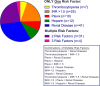
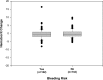
Measurements and main results: Thoracenteses were performed in 312 patients, 42% of whom had a risk for bleeding. Elevated INR, secondary to liver disease or warfarin, and renal disease were the two most common etiologies for bleeding risk, although many patients had multiple potential bleeding risks. There was no significant difference in pre- and postprocedural hematocrit levels in patients with a bleeding risk when compared with patients with no bleeding risk. No patient developed a hemothorax as a result of the thoracentesis.
Conclusions: This single-center, observational study suggests that thoracentesis may be safely performed without prior correction of coagulopathy, thrombocytopenia, or medication-induced bleeding risk. This may reduce the morbidity associated with transfusions or withholding of medications.
Figures
References
-
- Feller-Kopman D. Ultrasound-guided thoracentesis. Chest. 2006;129:1709–1714. - PubMed
-
- Feller-Kopman D. Therapeutic thoracentesis: the role of ultrasound and pleural manometry. Curr Opin Pulm Med. 2007;13:312–318. - PubMed
-
- Feller-Kopman D, Berkowitz D, Boiselle P, Ernst A. Large-volume thoracentesis and the risk of reexpansion pulmonary edema. Ann Thorac Surg. 2007;84:1656–1661. - PubMed
-
- Gordon CE, Feller-Kopman D, Balk EM, Smetana GW. Pneumothorax following thoracentesis: a systematic review and meta-analysis. Arch Intern Med. 2010;170:332–339. - PubMed
-
- Grogan DR, Irwin RS, Channick R, Raptopoulos V, Curley FJ, Bartter T, Corwin RW. Complications associated with thoracentesis: a prospective, randomized study comparing three different methods. Arch Intern Med. 1990;150:873–877. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

