Functional inositol 1,4,5-trisphosphate receptors assembled from concatenated homo- and heteromeric subunits
- PMID: 23955339
- PMCID: PMC3795275
- DOI: 10.1074/jbc.M113.502203
Functional inositol 1,4,5-trisphosphate receptors assembled from concatenated homo- and heteromeric subunits
Abstract
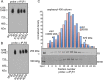
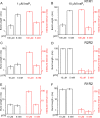
Vertebrate genomes code for three subtypes of inositol 1,4,5-trisphosphate (IP3) receptors (IP3R1, -2, and -3). Individual IP3R monomers are assembled to form homo- and heterotetrameric channels that mediate Ca(2+) release from intracellular stores. IP3R subtypes are regulated differentially by IP3, Ca(2+), ATP, and various other cellular factors and events. IP3R subtypes are seldom expressed in isolation in individual cell types, and cells often express different complements of IP3R subtypes. When multiple subtypes of IP3R are co-expressed, the subunit composition of channels cannot be specifically defined. Thus, how the subunit composition of heterotetrameric IP3R channels contributes to shaping the spatio-temporal properties of IP3-mediated Ca(2+) signals has been difficult to evaluate. To address this question, we created concatenated IP3R linked by short flexible linkers. Dimeric constructs were expressed in DT40-3KO cells, an IP3R null cell line. The dimeric proteins were localized to membranes, ran as intact dimeric proteins on SDS-PAGE, and migrated as an ∼1100-kDa band on blue native gels exactly as wild type IP3R. Importantly, IP3R channels formed from concatenated dimers were fully functional as indicated by agonist-induced Ca(2+) release. Using single channel "on-nucleus" patch clamp, the channels assembled from homodimers were essentially indistinguishable from those formed by the wild type receptor. However, the activity of channels formed from concatenated IP3R1 and IP3R2 heterodimers was dominated by IP3R2 in terms of the characteristics of regulation by ATP. These studies provide the first insight into the regulation of heterotetrameric IP3R of defined composition. Importantly, the results indicate that the properties of these channels are not simply a blend of those of the constituent IP3R monomers.
Keywords: ATP; Calcium Imaging; Calcium Intracellular Release; Calcium Signaling; Inositol 1,4,5-Trisphosphate Receptor; Signal Transduction; Single Channel Recording.
Figures







References
-
- Berridge M. J., Lipp P., Bootman M. D. (2000) The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 1, 11–21 - PubMed
-
- Nicotera P., Orrenius S. (1992) Ca2+ and cell death. Ann. N.Y. Acad. Sci. 648, 17–27 - PubMed
-
- Goonasekera S. A., Molkentin J. D. (2012) Unraveling the secrets of a double life: contractile versus signaling Ca2+ in a cardiac myocyte. J. Mol. Cell. Cardiol. 52, 317–322 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

