Randomized, placebo-controlled trial to assess the safety and immunogenicity of an adenovirus type 35-based circumsporozoite malaria vaccine in healthy adults
- PMID: 23955431
- PMCID: PMC4162066
- DOI: 10.4161/hv.26038
Randomized, placebo-controlled trial to assess the safety and immunogenicity of an adenovirus type 35-based circumsporozoite malaria vaccine in healthy adults
Abstract
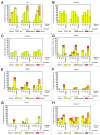
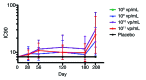
Malaria results in over 650,000 deaths each year; thus, there is an urgent need for an effective vaccine. Pre-clinical studies and recently reported human trials suggest that pre-erythrocytic stage vaccines can provide protection against infection. A Phase 1, randomized, placebo-controlled, dose-escalation study was conducted with a vaccine composed of a replication-deficient adenovirus-35 backbone with P. falciparum circumsporozoite (CS) surface antigen (Ad35.CS.01). Healthy adult subjects received three doses of 10 (8), 10 (9), 10 (10), or 10 (11) vp/mL Ad35.CS.01 vaccine or saline placebo intramuscularly at 0, 1, and 6-mo intervals. Adverse events were assessed and anti-CS antibody responses were determined by ELISA. Seventy-two individuals were enrolled, with age, gender, and ethnicity similar across each study arm. While the vaccine was generally well tolerated, adverse events were more frequent in the highest dose groups (10 (10) and 10 (11) vp/mL). More robust humoral responses were also noted at the highest doses, with 73% developing a positive ELISA response after the three dose series of 10 (11) vp/mL. The Ad35.CS.01 vaccine was most immunogenic at the highest dosages (10 (10) and 10 (11) vp/mL). Reactogenicity findings were more common after the 10 (11) vp/mL dose, although most were mild or moderate in nature and resolved without therapy.
Keywords: adenovirus; circumsporozoite; malaria; vaccine.
Figures

References
-
- World Health Organization. WHO Facts on Malaria. 2013.
-
- Mandell GL, Douglas RG, Bennett JE, Dolin R. Mandell, Douglas, and Bennett's principles and practice of infectious diseases 6th ed. Philadelphia: Elsevier Churchill Livingstone; 2005.
-
- John CC, Moormann AM, Pregibon DC, Sumba PO, McHugh MM, Narum DL, Lanar DE, Schluchter MD, Kazura JW. Correlation of high levels of antibodies to multiple pre-erythrocytic Plasmodium falciparum antigens and protection from infection. Am J Trop Med Hyg. 2005;73:222–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
