Regulation of Cell Proliferation by the Guanosine-Adenosine Mechanism: Role of Adenosine Receptors
- PMID: 23956837
- PMCID: PMC3743120
- DOI: 10.1002/phy2.24
Regulation of Cell Proliferation by the Guanosine-Adenosine Mechanism: Role of Adenosine Receptors
Abstract
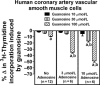
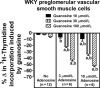
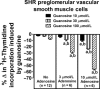
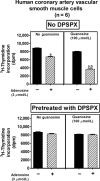
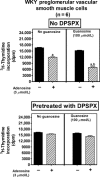
A recent study (American Journal of Physiology-Cell Physiology 304: C406-C421, 2013) suggests that extracellular guanosine increases extracellular adenosine by modifying the disposition of extracellular adenosine ("guanosine-adenosine mechanism") and that the guanosine-adenosine mechanism is not mediated by classical adenosine transport systems (SLC28 and SLC29 families) nor by classical adenosine-metabolizing enzymes. The present investigation had two aims: 1) to test the hypothesis that the "guanosine-adenosine mechanism" affects cell proliferation; and 2) to determine whether the transporters SLC19A1, SLC19A2, SLC19A3 or SLC22A2 (known to carrier guanosine analogues) might be responsible for the guanosine-adenosine mechanism. In the absence of added adenosine, guanosine had little effect on the proliferation of coronary artery vascular smooth muscle cells (vascular conduit cells) or preglomerular vascular smooth muscle cells (vascular resistance cells). However, in the presence of added adenosine (3 or 10 μmol/L), guanosine (10 to 100 μmol/L) decreased proliferation of both cell types, thus resulting in a highly significant (p<0.000001) interaction between guanosine and adenosine on cell proliferation. The guanosine-adenosine interaction on cell proliferation was abolished by 1,3-dipropyl-8-(p-sulfophenyl)xanthine (adenosine receptor antagonist). Guanosine (30 μmol/L) increased extracellular levels of adenosine when adenosine (3 μmol/L) was added to the medium. This effect was not reproduced by high concentrations of methotrexate (100 μmol/L), thiamine (1000 μmol/L), chloroquine (1000 μmol/L) or acyclovir (10,000 μmol/L), archetypal substrates for SLC19A1, SLC19A2, SLC19A3, and SLC22A2, respectively; and guanosine still increased adenosine levels in the presence of these compounds.
Conclusion: The guanosine-adenosine mechanism affects cell proliferation and is not mediated by SLC19A1, SLC19A2, SLC19A3 or SLC22A2.
Keywords: SLC19 family; SLC22A2; adenosine; cell proliferation; guanosine.
Figures


Similar articles
-
Guanosine regulates adenosine levels in the kidney.Physiol Rep. 2014 May 28;2(5):e12028. doi: 10.14814/phy2.12028. Print 2014 May 1. Physiol Rep. 2014. PMID: 24872359 Free PMC article.
-
Extracellular guanosine regulates extracellular adenosine levels.Am J Physiol Cell Physiol. 2013 Mar 1;304(5):C406-21. doi: 10.1152/ajpcell.00212.2012. Epub 2012 Dec 12. Am J Physiol Cell Physiol. 2013. PMID: 23242185 Free PMC article.
-
Cyclic AMP-adenosine pathway inhibits vascular smooth muscle cell growth.Hypertension. 1996 Nov;28(5):765-71. doi: 10.1161/01.hyp.28.5.765. Hypertension. 1996. PMID: 8901821
-
Folate and thiamine transporters mediated by facilitative carriers (SLC19A1-3 and SLC46A1) and folate receptors.Mol Aspects Med. 2013 Apr-Jun;34(2-3):373-85. doi: 10.1016/j.mam.2012.07.006. Mol Aspects Med. 2013. PMID: 23506878 Free PMC article. Review.
-
SLC19: the folate/thiamine transporter family.Pflugers Arch. 2004 Feb;447(5):641-6. doi: 10.1007/s00424-003-1068-1. Epub 2003 May 6. Pflugers Arch. 2004. PMID: 14770311 Review.
Cited by
-
8-Aminoguanine Induces Diuresis, Natriuresis, and Glucosuria by Inhibiting Purine Nucleoside Phosphorylase and Reduces Potassium Excretion by Inhibiting Rac1.J Am Heart Assoc. 2018 Nov 6;7(21):e010085. doi: 10.1161/JAHA.118.010085. J Am Heart Assoc. 2018. PMID: 30608204 Free PMC article.
-
Adenosine Attenuates Human Coronary Artery Smooth Muscle Cell Proliferation by Inhibiting Multiple Signaling Pathways That Converge on Cyclin D.Hypertension. 2015 Dec;66(6):1207-19. doi: 10.1161/HYPERTENSIONAHA.115.05912. Epub 2015 Sep 28. Hypertension. 2015. PMID: 26416848 Free PMC article.
-
Guanosine inhibits LPS-induced pro-inflammatory response and oxidative stress in hippocampal astrocytes through the heme oxygenase-1 pathway.Purinergic Signal. 2015 Dec;11(4):571-80. doi: 10.1007/s11302-015-9475-2. Epub 2015 Oct 2. Purinergic Signal. 2015. PMID: 26431832 Free PMC article.
-
Guanosine promotes cytotoxicity via adenosine receptors and induces apoptosis in temozolomide-treated A172 glioma cells.Purinergic Signal. 2017 Sep;13(3):305-318. doi: 10.1007/s11302-017-9562-7. Epub 2017 May 23. Purinergic Signal. 2017. PMID: 28536931 Free PMC article.
-
Guanosine regulates adenosine levels in the kidney.Physiol Rep. 2014 May 28;2(5):e12028. doi: 10.14814/phy2.12028. Print 2014 May 1. Physiol Rep. 2014. PMID: 24872359 Free PMC article.
References
-
- Andresen BT, Gillespie DG, Mi Z, Dubey RK, Jackson EK. Role of adenosine A1 receptors in modulating extracellular adenosine levels. J. Pharmacol. Exp. Ther. 1999;291:76–80. - PubMed
-
- Bours MJL, Swennen ELR, Cronstein F, Di Virgilio BN, Dagnelie PC. Adenosine 5′-triphosphate and adenosine as endogenous signaling molecules in immunity and inflammation. Pharmacol. Ther. 2006;112:358–404. - PubMed
-
- Campbell JH, Tachas G, Black MJ, Cockerill G, Campbell GR. Molecular biology of vascular hypertrophy. Basic Res. Cardiol. 1991;86(Suppl. 1):3–11. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

