Redox-neutral α-C-H bond functionalization of secondary amines with concurrent C-P bond formation/N-alkylation
- PMID: 23957378
- PMCID: PMC3818705
- DOI: 10.1021/ol401858k
Redox-neutral α-C-H bond functionalization of secondary amines with concurrent C-P bond formation/N-alkylation
Abstract
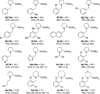
Redox-neutral formation of C-P bonds in the α-position of amines was achieved via a process that features a combination of an oxidative α-C-H bond functionalization and a reductive N-alkylation. Benzoic acid functions as an efficient catalyst in this three-component reaction of cyclic secondary amines, aldehydes and phosphine oxides to provide rapid access to α-amino phosphine oxides not easily accessible by classic Kabachnik-Fields reactions.
Figures
References
-
- Kukhar VP, Hudson HR, editors. Aminophosphonic and Aminophosphinic Acids: Chemistry and Biological Activity. Chichester: John Wiley & Sons; 2000.
-
- Lejczak B, Kafarski P. Top. Heterocycl. Chem. 2009;20:31.
- Orsini F, Sello G, Sisti M. Curr. Med. Chem. 2010;17:264. - PubMed
- Naydenova ED, Todorov PT, Troev KD. Amino Acids. 2010;38:23. - PubMed
- Mucha A, Kafarski P, Berlicki L. J. Med. Chem. 2011;54:5955. - PubMed
- Bhattacharya AK, Rana KC, Pannecouque C, De Clercq E. Chem Med Chem. 2012;7:1601. - PubMed
-
-
Recent reviews: Ordóñez M, Rojas-Cabrera H, Cativiela C. Tetrahedron. 2009;65:17. Kudzina ZH, Kudzinb MH, Drabowiczc J, Stevens CV. Curr. Org. Chem. 2011;15:2015. Liu BJ, Cen CC, Wu MS, Kong DL. Asian. J. Chem. 2011;23:1417. Ordóñez M, Viveros-Ceballos JL, Cativiela C, Arizpe A. Curr. Org. Synth. 2012;9:310. Ordóñez M, Sayago FJ, Cativiela C. Tetrahedron. 2012;68:6369.
-
-
- Kabachnik MI, Medved TY. Dokl. Akad. Nauk SSSR. 1952;83:689.
- Fields EK. J. Am. Chem. Soc. 1952;74:1528.
-
-
Selected reviews: Zefirov NS, Matveeva ED. ARKIVOC. 2008:1. Keglevich G, Balint E. Molecules. 2012;17:12821.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous