Site-specific PEGylation enhances the pharmacokinetic properties and antitumor activity of interferon beta-1b
- PMID: 23962003
- PMCID: PMC3868373
- DOI: 10.1089/jir.2012.0148
Site-specific PEGylation enhances the pharmacokinetic properties and antitumor activity of interferon beta-1b
Abstract
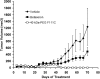
Interferon beta (IFN-β) is widely used to ameliorate disease progression in patients with Multiple Sclerosis. IFN-β has a short half-life in humans, necessitating frequent administration for optimum effectiveness. Covalent modification of IFN-β with polyethylene glycol (PEG) improves the pharmacokinetic properties of the protein, but can adversely affect the protein's in vitro bioactivity. Random modification of lysine residues in IFN-β with amine-reactive PEGs decreased the in vitro bioactivity of the protein 50-fold, presumably due to modification of lysine residues near critical receptor binding sites. PEGylated IFN-β proteins that retained high in vitro bioactivity could be obtained by selective modification of the N-terminus of the protein with PEG. Here we use site-specific PEGylation technology (targeted attachment of a cysteine-reactive-PEG to an engineered cysteine residue in IFN-β) to identify several additional amino acid positions where PEG can be attached to IFN-β without appreciable loss of in vitro bioactivity. Unexpectedly, we found that most of the PEG-IFN-β analogs showed 11- to 78-fold improved in vitro bioactivities relative to their unPEGylated parent proteins and to IFN-β-1b. In vivo studies showed that a lead PEG-IFN-β protein had improved pharmacokinetic properties compared to IFN-β and was significantly more effective than IFN-β at inhibiting growth of a human tumor xenograft in athymic mice.
Figures

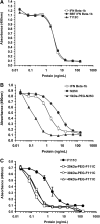
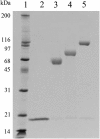
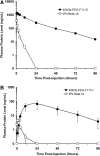
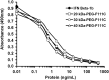
References
-
- Abuchowski A, Kazo GM, Verhoest CR, van Es T, Kafkewitz D, Nucci ML, Viau AT, Davis FF. 1984. Cancer therapy with chemically modified enzymes. I. Antitumor properties of polyethylene glycol-asparaginase conjugates. Cancer Biochem Biophys 7:175–186 - PubMed
-
- Bailon P, Palleroni A, Schaffer CA, Spence CL, Fung W-J, Porter JE, Ehrlich GK, Pan W, Xu Z-X, Modi MW, Farid A, Berthold W, Graves M. 2001. Rational design of a potent, long-lasting form of interferon: a 40 kDa branched polyethylene glycol-conjugated interferon-α2a for the treatment of hepatitis C. Bioconjug Chem 12:195–202 - PubMed
-
- Baker DP, Lin EY, Lin K, Pellegrini M, Petter RC, Chen LL, Arduini RM, Brickelmaier M, Wen D, Hess DM, Chen L, Grant D, Whitty A, Gill A, Lindner DJ, Pepinsky RB. 2006. N-terminally PEGylated human interferon-beta-1a with improved pharmacokinetic properties and in vivo efficacy in a melanoma angiogenesis model. Bioconjug Chem 17:179–188 - PubMed
-
- Basu A, Yang K, Wang M, Liu S, Chintala R, Palm T, Zhao H, Peng P, Wu D, Zhang Z, Hua J, Hsieh MC, Zhou J, Petti G, Li X, Janjua A, Mendez M, Liu J, Longley C, Zhang Z, Mehlig M, Borowski V, Viswanathan M, Filpula D. 2006. Structure-function engineering of interferon-beta-1b for improved stability, solubility, potency, immunogenicity, and pharmacokinetic properties by site-selective monoPEGylation. Bioconjug Chem 17:618–630 - PubMed
-
- Bell SJ, Fam CM, Chlipala EA, Carlson SJ, Lee JI, Rosendahl MS, Doherty DH, Cox GN. 2008. Enhanced circulating half-life and anti-tumor activity of a site-specific pegylated interferon - α protein therapeutic. Bioconjug Chem 19:299–305 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

