Conformational changes represent the rate-limiting step in the transport cycle of maize sucrose transporter1
- PMID: 23964025
- PMCID: PMC3784595
- DOI: 10.1105/tpc.113.113621
Conformational changes represent the rate-limiting step in the transport cycle of maize sucrose transporter1
Abstract
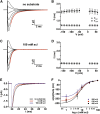
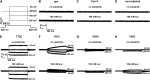
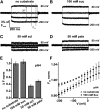
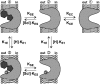
Proton-driven Suc transporters allow phloem cells of higher plants to accumulate Suc to more than 1 M, which is up to ~1000-fold higher than in the surrounding extracellular space. The carrier protein can accomplish this task only because proton and Suc transport are tightly coupled. This study provides insights into this coupling by resolving the first step in the transport cycle of the Suc transporter SUT1 from maize (Zea mays). Voltage clamp fluorometry measurements combining electrophysiological techniques with fluorescence-based methods enable the visualization of conformational changes of SUT1 expressed in Xenopus laevis oocytes. Using the Suc derivate sucralose, binding of which hinders conformational changes of SUT1, the association of protons to the carrier could be dissected from transport-associated movements of the protein. These combined approaches enabled us to resolve the binding of protons to the carrier and its interrelationship with the alternating movement of the protein. The data indicate that the rate-limiting step of the reaction cycle is determined by the accessibility of the proton binding site. This, in turn, is determined by the conformational change of the SUT1 protein, alternately exposing the binding pockets to the inward and to the outward face of the membrane.
Figures

Similar articles
-
Sucrose- and H-dependent charge movements associated with the gating of sucrose transporter ZmSUT1.PLoS One. 2010 Sep 7;5(9):e12605. doi: 10.1371/journal.pone.0012605. PLoS One. 2010. PMID: 20838661 Free PMC article.
-
Phloem-localized, proton-coupled sucrose carrier ZmSUT1 mediates sucrose efflux under the control of the sucrose gradient and the proton motive force.J Biol Chem. 2005 Jun 3;280(22):21437-43. doi: 10.1074/jbc.M501785200. Epub 2005 Apr 1. J Biol Chem. 2005. PMID: 15805107
-
Sucrose Transporter ZmSut1 Expression and Localization Uncover New Insights into Sucrose Phloem Loading.Plant Physiol. 2016 Nov;172(3):1876-1898. doi: 10.1104/pp.16.00884. Epub 2016 Sep 12. Plant Physiol. 2016. PMID: 27621426 Free PMC article.
-
Sucrose transport in higher plants.Int Rev Cytol. 1998;178:41-71. doi: 10.1016/s0074-7696(08)62135-x. Int Rev Cytol. 1998. PMID: 9348668 Review.
-
Plant sucrose transporters from a biophysical point of view.Mol Plant. 2011 May;4(3):395-406. doi: 10.1093/mp/ssr029. Epub 2011 Apr 18. Mol Plant. 2011. PMID: 21502662 Review.
Cited by
-
Current Methods to Unravel the Functional Properties of Lysosomal Ion Channels and Transporters.Cells. 2022 Mar 8;11(6):921. doi: 10.3390/cells11060921. Cells. 2022. PMID: 35326372 Free PMC article. Review.
-
The Surprising Dynamics of Electrochemical Coupling at Membrane Sandwiches in Plants.Plants (Basel). 2023 Jan 3;12(1):204. doi: 10.3390/plants12010204. Plants (Basel). 2023. PMID: 36616332 Free PMC article.
-
Plant glucose transporter structure and function.Pflugers Arch. 2020 Sep;472(9):1111-1128. doi: 10.1007/s00424-020-02449-3. Epub 2020 Aug 26. Pflugers Arch. 2020. PMID: 32845347 Free PMC article. Review.
-
Electrophysiology and fluorescence to investigate cation channels and transporters in isolated plant vacuoles.Stress Biol. 2022 Oct 1;2(1):42. doi: 10.1007/s44154-022-00064-z. Stress Biol. 2022. PMID: 37676514 Free PMC article. Review.
-
Overexpression of a Grapevine Sucrose Transporter (VvSUC27) in Tobacco Improves Plant Growth Rate in the Presence of Sucrose In vitro.Front Plant Sci. 2017 Jun 20;8:1069. doi: 10.3389/fpls.2017.01069. eCollection 2017. Front Plant Sci. 2017. PMID: 28676814 Free PMC article.
References
-
- Abramson J., Smirnova I., Kasho V., Verner G., Kaback H.R., Iwata S. (2003). Structure and mechanism of the lactose permease of Escherichia coli. Science 301: 610–615 - PubMed
-
- Aoki N., Hirose T., Scofield G.N., Whitfeld P.R., Furbank R.T. (2003). The sucrose transporter gene family in rice. Plant Cell Physiol. 44: 223–232 - PubMed
-
- Aoki N., Hirose T., Takahashi S., Ono K., Ishimaru K., Ohsugi R. (1999). Molecular cloning and expression analysis of a gene for a sucrose transporter in maize (Zea mays L.). Plant Cell Physiol. 40: 1072–1078 - PubMed
-
- Boorer K.J., Frommer W.B., Bush D.R., Kreman M., Loo D.D., Wright E.M. (1996b). Kinetics and specificity of a H+/amino acid transporter from Arabidopsis thaliana. J. Biol. Chem. 271: 2213–2220 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources

