Atg16l1 is required for autophagy in intestinal epithelial cells and protection of mice from Salmonella infection
- PMID: 23973919
- PMCID: PMC3840157
- DOI: 10.1053/j.gastro.2013.08.035
Atg16l1 is required for autophagy in intestinal epithelial cells and protection of mice from Salmonella infection
Abstract
Background & aims: Intestinal epithelial cells aid in mucosal defense by providing a physical barrier against entry of pathogenic bacteria and secreting antimicrobial peptides (AMPs). Autophagy is an important component of immune homeostasis. However, little is known about its role in specific cell types during bacterial infection in vivo. We investigated the role of autophagy in the response of intestinal epithelial and antigen-presenting cells to Salmonella infection in mice.
Methods: We generated mice deficient in Atg16l1 in epithelial cells (Atg16l1(f/f) × Villin-cre) or CD11c(+) cells (Atg16l1(f/f) × CD11c-cre); these mice were used to assess cell type-specific antibacterial autophagy. All responses were compared with Atg16l1(f/f) mice (controls). Mice were infected with Salmonella enterica serovar typhimurium; cecum and small-intestine tissues were collected for immunofluorescence, histology, and quantitative reverse-transcription polymerase chain reaction analyses of cytokines and AMPs. Modulators of autophagy were screened to evaluate their effects on antibacterial responses in human epithelial cells.
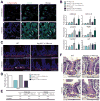
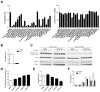
Results: Autophagy was induced in small intestine and cecum after infection with S typhimurium, and required Atg16l1. S typhimurium colocalized with microtubule-associated protein 1 light chain 3β (Map1lc3b or LC3) in the intestinal epithelium of control mice but not in Atg16l1(f/f) × Villin-cre mice. Atg16l1(f/f) × Villin-cre mice also had fewer Paneth cells and abnormal granule morphology, leading to reduced expression of AMPs. Consistent with these defective immune responses, Atg16l1(f/f) × Villin-cre mice had increased inflammation and systemic translocation of bacteria compared with control mice. In contrast, we observed few differences between Atg16l1(f/f) × CD11c-cre and control mice. Trifluoperazine promoted autophagy and bacterial clearance in HeLa cells; these effects were reduced upon knockdown of ATG16L1.
Conclusions: Atg16l1 regulates autophagy in intestinal epithelial cells and is required for bacterial clearance. It also is required to prevent systemic infection of mice with enteric bacteria.
Keywords: AMP; ATG; Autophagy; Discosoma sp. red fluorescent protein; EGFP; FAE; GFP; IL; Intestinal Barrier; LC3; MEF; MLN; Map1lc3b; Mouse Model; Mucosa; SCV; Salmonella-containing vacuole; TFP; antimicrobial peptide; autophagy-related gene; dsRed; enhanced green fluorescent protein; follicle-associated epithelium; green fluorescent protein; interleukin; light chain 3; mRNA; mesenteric lymph nodes; messenger RNA; microtubule-associated protein 1 light chain 3b; mouse embryonic fibroblast; trifluoperazine.
Copyright © 2013 AGA Institute. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures




References
-
- Weidberg H, Shvets E, Elazar Z. Biogenesis and cargo selectivity of autophagosomes. Annu Rev Biochem. 2011;80:125–56. - PubMed
-
- Kuballa P, Nolte WM, Castoreno AB, et al. Autophagy and the immune system. Annu Rev Immunol. 2012;30:611–46. - PubMed
-
- Kim JJ, Lee HM, Shin DM, et al. Host cell autophagy activated by antibiotics is required for their effective antimycobacterial drug action. Cell Host Microbe. 2012;11:457–68. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P30 DK040561/DK/NIDDK NIH HHS/United States
- R01 DK060049/DK/NIDDK NIH HHS/United States
- DK033506/DK/NIDDK NIH HHS/United States
- P30 DK043351/DK/NIDDK NIH HHS/United States
- R01 DK092405/DK/NIDDK NIH HHS/United States
- DK082427/DK/NIDDK NIH HHS/United States
- R56 AI093588/AI/NIAID NIH HHS/United States
- DK097485/DK/NIDDK NIH HHS/United States
- R01 AI084887/AI/NIAID NIH HHS/United States
- DK043351/DK/NIDDK NIH HHS/United States
- P01 DK033506/DK/NIDDK NIH HHS/United States
- R01 DK068181/DK/NIDDK NIH HHS/United States
- K99 AG045144/AG/NIA NIH HHS/United States
- DK092405/DK/NIDDK NIH HHS/United States
- DK068181/DK/NIDDK NIH HHS/United States
- R01 AI054483/AI/NIAID NIH HHS/United States
- AI093588/AI/NIAID NIH HHS/United States
- R00 AG045144/AG/NIA NIH HHS/United States
- AI054483/AI/NIAID NIH HHS/United States
- DK060049/DK/NIDDK NIH HHS/United States
- R01 DK082427/DK/NIDDK NIH HHS/United States
- R01 DK097485/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

