A disinhibitory microcircuit initiates critical-period plasticity in the visual cortex
- PMID: 23975100
- PMCID: PMC3962838
- DOI: 10.1038/nature12485
A disinhibitory microcircuit initiates critical-period plasticity in the visual cortex
Abstract
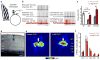
Early sensory experience instructs the maturation of neural circuitry in the cortex. This has been studied extensively in the primary visual cortex, in which loss of vision to one eye permanently degrades cortical responsiveness to that eye, a phenomenon known as ocular dominance plasticity (ODP). Cortical inhibition mediates this process, but the precise role of specific classes of inhibitory neurons in ODP is controversial. Here we report that evoked firing rates of binocular excitatory neurons in the primary visual cortex immediately drop by half when vision is restricted to one eye, but gradually return to normal over the following twenty-four hours, despite the fact that vision remains restricted to one eye. This restoration of binocular-like excitatory firing rates after monocular deprivation results from a rapid, although transient, reduction in the firing rates of fast-spiking, parvalbumin-positive (PV) interneurons, which in turn can be attributed to a decrease in local excitatory circuit input onto PV interneurons. This reduction in PV-cell-evoked responses after monocular lid suture is restricted to the critical period for ODP and appears to be necessary for subsequent shifts in excitatory ODP. Pharmacologically enhancing inhibition at the time of sight deprivation blocks ODP and, conversely, pharmacogenetic reduction of PV cell firing rates can extend the critical period for ODP. These findings define the microcircuit changes initiating competitive plasticity during critical periods of cortical development. Moreover, they show that the restoration of evoked firing rates of layer 2/3 pyramidal neurons by PV-specific disinhibition is a key step in the progression of ODP.
Figures


References
-
- White LE, Fitzpatrick D. Vision and cortical map development. Neuron. 2007;56:327–338. - PubMed
-
- Wiesel TN, Hubel DH. Single-Cell Responses in Striate Cortex of Kittens Deprived of Vision in One Eye. J Neurophysiol. 1963;26:1003–1017. - PubMed
-
- Levelt CN, Hubener M. Critical-period plasticity in the visual cortex. Annu Rev Neurosci. 2012;35:309–330. - PubMed
-
- Hensch TK. Critical period plasticity in local cortical circuits. Nat Rev Neurosci. 2005;6:877–888. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

