The clathrin adaptor complex AP-2 mediates endocytosis of brassinosteroid insensitive1 in Arabidopsis
- PMID: 23975899
- PMCID: PMC3784593
- DOI: 10.1105/tpc.113.114058
The clathrin adaptor complex AP-2 mediates endocytosis of brassinosteroid insensitive1 in Arabidopsis
Abstract
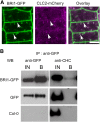
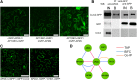
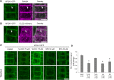
Clathrin-mediated endocytosis (CME) regulates many aspects of plant development, including hormone signaling and responses to environmental stresses. Despite the importance of this process, the machinery that regulates CME in plants is largely unknown. In mammals, the heterotetrameric adaptor protein complex-2 (AP-2) is required for the formation of clathrin-coated vesicles at the plasma membrane (PM). Although the existence of AP-2 has been predicted in Arabidopsis thaliana, the biochemistry and functionality of the complex is still uncharacterized. Here, we identified all the subunits of the Arabidopsis AP-2 by tandem affinity purification and found that one of the large AP-2 subunits, AP2A1, localized at the PM and interacted with clathrin. Furthermore, endocytosis of the leucine-rich repeat receptor kinase, brassinosteroid insensitive1 (BRI1), was shown to depend on AP-2. Knockdown of the two Arabidopsis AP2A genes or overexpression of a dominant-negative version of the medium AP-2 subunit, AP2M, impaired BRI1 endocytosis and enhanced the brassinosteroid signaling. Our data reveal that the CME machinery in Arabidopsis is evolutionarily conserved and that AP-2 functions in receptor-mediated endocytosis.
Figures


References
-
- Abas L., Benjamins R., Malenica N., Paciorek T., Wiśniewska J., Moulinier-Anzola J.C., Sieberer T., Friml J., Luschnig C. (2006). Intracellular trafficking and proteolysis of the Arabidopsis auxin-efflux facilitator PIN2 are involved in root gravitropism. Nat. Cell Biol. 8: 249–256 Erratum. Nat. Cell Biol. 8, 424. - PubMed
-
- Adam T., Bouhidel K., Der C., Robert F., Najid A., Simon-Plas F., Leborgne-Castel N. (2012). Constitutive expression of clathrin hub hinders elicitor-induced clathrin-mediated endocytosis and defense gene expression in plant cells. FEBS Lett. 586: 3293–3298 - PubMed
-
- Bar M., Avni A. (2009). EHD2 inhibits ligand-induced endocytosis and signaling of the leucine-rich repeat receptor-like protein LeEix2. Plant J. 59: 600–611 - PubMed
-
- Barth M., Holstein S.E.H. (2004). Identification and functional characterization of Arabidopsis AP180, a binding partner of plant αC-adaptin. J. Cell Sci. 117: 2051–2062 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

