Tacrolimus decreases albuminuria in patients with IgA nephropathy and normal blood pressure: a double-blind randomized controlled trial of efficacy of tacrolimus on IgA nephropathy
- PMID: 23977072
- PMCID: PMC3747236
- DOI: 10.1371/journal.pone.0071545
Tacrolimus decreases albuminuria in patients with IgA nephropathy and normal blood pressure: a double-blind randomized controlled trial of efficacy of tacrolimus on IgA nephropathy
Abstract
Background: Treatment remains uncertain for IgA nephropathy patients with mild to moderate proteinuria, for whom anti-hypertensive medication or the RAS blocker is not applicable due to low blood pressure.
Trial design: A double blinded randomized trial.
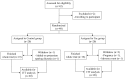
Methods: The anti-proteinuric effect of tacrolimus was explored for 40 biopsy-proven mild IgA nephropathies for 16 weeks. We randomly assigned patients either to receive tacrolimus or placebo with stratification by using a renin angiotensin system blocker. The primary outcome was the percentage change of final UACR compared to the baseline value (pcUACR).
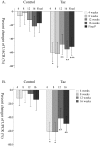
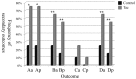
Results: The mean value of pcUACR at 12-week and 16-week visits (primary outcome) was decreased more in the Tac group compared to the control group (-52.0±26.4 vs -17.3±29.3%, p = 0.001). At each visit, pcUACR was also decreased more in the Tac group compared to the control group. In the Tac group, the pcUACRs were -60.2±28.2%, -62.2±33.9%, -48.5±29.8%, and -55.5±24.0%, and, in the control group, -6.8±32.2%, -2.5±35.9%, -12.7±34.2%, and -21.9±30.6%, at 4-week, 8-week, 12-week, and 16-week visits, respectively. The pre-defined secondary outcomes were better in the Tac group compared to the control group. The frequency of decrease in pcUACR and percentage change of UPCR (pcUPCR) ≥50% at 16 weeks were 65.0% (13/20) and 55.0% (11/20)in the Tac group, and 25.0% (5/20) and 15.0% (3/20), in the control group, respectively (p = 0.025 for pcUACR and p = 0.019 for pcUPCR). However, tacrolimus wasn't effective with a dose of 0.05 mg/kg/day in patients taking ARB. The adverse events were tolerable.
Conclusion: Tacrolimus effectively reduced proteinuria in IgA nephropathy with normal blood pressure. This suggested that tacrolimus could be an alternative to corticosteroid and RAS blocker for IgA nephropathy patients who cannot endure anti-hypertensive medication.
Trial registration: Clinicaltrial.gov NCT1224028.
Trial registration: ClinicalTrials.gov NCT01224028 NCT01224028.
Conflict of interest statement
Figures
Similar articles
-
Short-term anti-proteinuric effect of tacrolimus is not related to preservation of the glomerular filtration rate in IgA nephropathy: A 5-year follow-up study.PLoS One. 2017 Nov 20;12(11):e0188375. doi: 10.1371/journal.pone.0188375. eCollection 2017. PLoS One. 2017. PMID: 29155873 Free PMC article. Clinical Trial.
-
Targeted-release budesonide versus placebo in patients with IgA nephropathy (NEFIGAN): a double-blind, randomised, placebo-controlled phase 2b trial.Lancet. 2017 May 27;389(10084):2117-2127. doi: 10.1016/S0140-6736(17)30550-0. Epub 2017 Mar 28. Lancet. 2017. PMID: 28363480 Clinical Trial.
-
Hong Kong study using valsartan in IgA nephropathy (HKVIN): a double-blind, randomized, placebo-controlled study.Am J Kidney Dis. 2006 May;47(5):751-60. doi: 10.1053/j.ajkd.2006.01.017. Am J Kidney Dis. 2006. PMID: 16632013 Clinical Trial.
-
Treatment of IgA nephropathy.J Nephrol. 2016 Feb;29(1):21-5. doi: 10.1007/s40620-015-0248-3. Epub 2015 Nov 17. J Nephrol. 2016. PMID: 26577268 Review.
-
ACEI/ARB therapy for IgA nephropathy: a meta analysis of randomised controlled trials.Int J Clin Pract. 2009 Jun;63(6):880-8. doi: 10.1111/j.1742-1241.2009.02038.x. Int J Clin Pract. 2009. PMID: 19490198 Review.
Cited by
-
Efficacy and safety of calcineurin inhibitor treatment for IgA nephropathy: a meta-analysis.BMC Nephrol. 2017 Feb 13;18(1):61. doi: 10.1186/s12882-017-0467-z. BMC Nephrol. 2017. PMID: 28193247 Free PMC article. Review.
-
Navigating Adult-Onset IgA Vasculitis-Associated Nephritis.Life (Basel). 2024 Jul 25;14(8):930. doi: 10.3390/life14080930. Life (Basel). 2024. PMID: 39202674 Free PMC article. Review.
-
Has The Time Arrived to Refine The Indications of Immunosuppressive Therapy and Prognosis in IgA Nephropathy?J Clin Med. 2019 Oct 2;8(10):1584. doi: 10.3390/jcm8101584. J Clin Med. 2019. PMID: 31581654 Free PMC article. Review.
-
The efficacy and safety of immunosuppressive therapies in the treatment of IgA nephropathy: A network meta-analysis.Sci Rep. 2020 Apr 8;10(1):6062. doi: 10.1038/s41598-020-63170-w. Sci Rep. 2020. PMID: 32269271 Free PMC article.
-
Comparison of steroid-pulse therapy and combined with mizoribine in IgA nephropathy: a randomized controlled trial.Clin Exp Nephrol. 2016 Dec;20(6):896-903. doi: 10.1007/s10157-016-1226-3. Epub 2016 Jan 13. Clin Exp Nephrol. 2016. PMID: 26758039 Clinical Trial.
References
-
- Chang JH, Kim DK, Kim HW, Park SY, Yoo TH, et al. (2009) Changing prevalence of glomerular diseases in Korean adults: a review of 20 years of experience. Nephrol Dial Transplant 24(8): 2406–2410. - PubMed
-
- Lee H, Kim DK, Oh KH, Joo KW, Kim YS, et al. (2013) Mortality and renal outcome of primary glomerulonephritis in Korea: Observation in 1,943 biopsied cases. Am J Nephrol 37(1): 74–83. - PubMed
-
- D'Amico G (1987) The commonest glomerulonephritis in the world: IgA nephropathy. Q J Med 64(245): 709–727. - PubMed
-
- Geddes CC, Rauta V, Gronhagen-Riska C, Bartosik LP, Jardine AG, et al. (2003) A tricontinental view of IgA nephropathy. Nephrol Dial Transplant 18(8): 1541–1548. - PubMed
-
- Floege J, Eitner F (2011) Current therapy for IgA nephropathy. J Am Soc Nephrol 22(10): 1785–1794. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

