A portable reverse transcription recombinase polymerase amplification assay for rapid detection of foot-and-mouth disease virus
- PMID: 23977101
- PMCID: PMC3748043
- DOI: 10.1371/journal.pone.0071642
A portable reverse transcription recombinase polymerase amplification assay for rapid detection of foot-and-mouth disease virus
Abstract
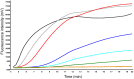
Foot-and-mouth disease (FMD) is a trans-boundary viral disease of livestock, which causes huge economic losses and constitutes a serious infectious threat for livestock farming worldwide. Early diagnosis of FMD helps to diminish its impact by adequate outbreak management. In this study, we describe the development of a real-time reverse transcription recombinase polymerase amplification (RT-RPA) assay for the detection of FMD virus (FMDV). The FMDV RT-RPA design targeted the 3D gene of FMDV and a 260 nt molecular RNA standard was used for assay validation. The RT-RPA assay was fast (4-10 minutes) and the analytical sensitivity was determined at 1436 RNA molecules detected by probit regression analysis. The FMDV RT-RPA assay detected RNA prepared from all seven FMDV serotypes but did not detect classical swine fever virus or swine vesicular disease virus. The FMDV RT-RPA assay was used in the field during the recent FMD outbreak in Egypt. In clinical samples, reverse transcription polymerase chain reaction (RT-PCR) and RT-RPA showed a diagnostic sensitivity of 100% and 98%, respectively. In conclusion, FMDV RT-RPA was quicker and much easier to handle in the field than real-time RT-PCR. Thus RT-RPA could be easily implemented to perform diagnostics at quarantine stations or farms for rapid spot-of-infection detection.
Conflict of interest statement
Figures

References
-
- Knowles NJ, Samuel AR (2003) Molecular epidemiology of foot-and-mouth disease virus. Virus research 91: 65–80. - PubMed
-
- Boothroyd JC, Highfield PE, Cross GA, Rowlands DJ, Lowe PA, et al. (1981) Molecular cloning of foot and mouth disease virus genome and nucleotide sequences in the structural protein genes. Nature 290: 800–802. - PubMed
-
- Rodriguez LL, Gay CG (2011) Development of vaccines toward the global control and eradication of foot-and-mouth disease. Expert review of vaccines 10: 377–387. - PubMed
-
- Ghoneim NH, Abdel-Karim AK, El-Shehawy L, Abdel-Moein KA (2010) Foot and mouth disease in animals in Sharkia governorate - Egypt. Transboundary and emerging diseases 57: 19–21. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

