S100A8 and S100A9 induce cytokine expression and regulate the NLRP3 inflammasome via ROS-dependent activation of NF-κB(1.)
- PMID: 23977231
- PMCID: PMC3747084
- DOI: 10.1371/journal.pone.0072138
S100A8 and S100A9 induce cytokine expression and regulate the NLRP3 inflammasome via ROS-dependent activation of NF-κB(1.)
Abstract
S100A8 and S100A9 are cytoplasmic proteins expressed by phagocytes. High concentrations of these proteins have been correlated with various inflammatory conditions, including autoimmune diseases such as rheumatoid arthritis and Crohn's disease, as well as autoinflammatory diseases. In the present study, we examined the effects of S100A8 and S100A9 on the secretion of cytokines and chemokines from PBMCs. S100A8 and S100A9 induced the secretion of cytokines such as IL-6, IL-8, and IL-1β. This secretion was associated with the activation and translocation of the transcription factor NF-κB. Inhibition studies using antisense RNA and the pharmacological agent BAY-117082 confirmed the involvement of NF-κB in IL-6, IL-8, and IL-1β secretion. S100A8- and S100A9-mediated activation of NF-κB, the NLR family, pyrin domain-containing 3 (NLRP3) protein, and pro-IL-1β expression was dependent on the generation of reactive oxygen species. This effect was synergistically enhanced by ATP, a known inflammasome activator. These results suggest that S100A8 and S100A9 enhance the inflammatory response by inducing cytokine secretion of PBMCs.
Conflict of interest statement
Figures

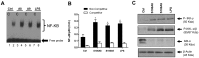
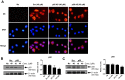
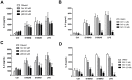
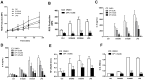
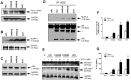
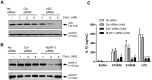
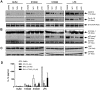
References
-
- Matzinger P (1994) Tolerance, danger, and the extended family. Annu Rev Immunol 12: 991–1045. - PubMed
-
- Bianchi ME (2007) DAMPs, PAMPs and alarmins: all we need to know about danger. J Leukoc Biol 81: 1–5. - PubMed
-
- Kessel C, Holzinger D, Foell D (2012) Phagocyte-derived S100 proteins in autoinflammation: Putative role in pathogenesis and usefulness as biomarkers. Clin Immunol. - PubMed
-
- Lim SY, Raftery MJ, Goyette J, Hsu K, Geczy CL (2009) Oxidative modifications of S100 proteins: functional regulation by redox. J Leukoc Biol 86: 577–587. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

